Consider the separation of inorganic and organic anions in Figure 26-7. a. What is the probable charge
Question:
Consider the separation of inorganic and organic anions in Figure 26-7.
a. What is the probable charge (Xn-) (Xn-)of pyruvate (peak 1010), 2-2-oxovalerate (peak 1616), and maleate (peak 2828)?
b. Iodide (peak 44 44) is a -1-1 ion. Explain its strong retention.
Figure 26-7
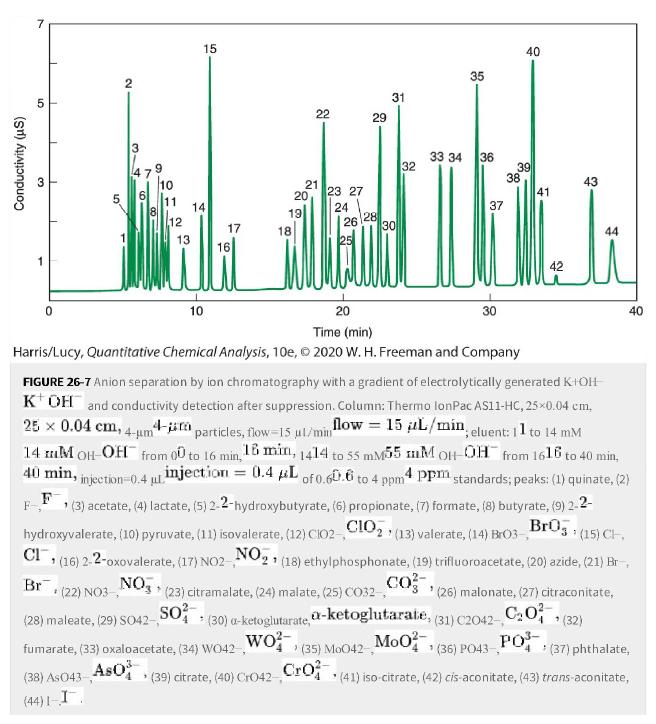
Transcribed Image Text:
Conductivity (us) 31 3 T 50 2 3 A 6 9 10 11 15 12 17 ili 10 19 18 20 22 21 23 27 24 26 29 28 31 . (39) citrate, (40) Cro42 Cro. 30 32 (35) Mo042- 33 34 35 36 37 1 30 20 Time (min) Harris/Lucy, Quantitative Chemical Analysis, 10e, © 2020 W. H. Freeman and Company 39 38 40 41 43 44 FIGURE 26-7 Anion separation by ion chromatography with a gradient of electrolytically generated K+OH KOH and conductivity detection after suppression. Column: Thermo lonPac AS11-HC, 25×0.04 cm, 25 x 0.04 cm, 4-um4 4- particles, flow-15 µl/min flow = 15 L/min, eluent: 11 to 14 mM 14 M OH-OH from 0 to 16 min, 18 min, 1414 to 55 mM55 mM OH-OH from 1616 to 40 min, 40 min, injection 0.4 injection = 0.4 μL of 0.60.6 to 4 ppm4 ppm standards; peaks: (1) quinate, (2) (3) acetate, (4) lactate, (5) 2-2-hydroxybutyrate, (6) propionate, (7) formate, (8) butyrate, (9) 2-2- F hydroxyvalerate, (10) pyruvate, (11) isovalerate, (12) C102 CIO₂, (13) valerate, (14) Bro3_BrO (15) CH CI, (16) 2-2-oxovalerate, (17) NO2 NO₂ (18) ethylphosphonate, (19) trifluoroacetate, (20) azide, (21) Br NO (23) citramalate, (24) malate, (25) CO32- CO (30) a-ketoglutarate, a-ketoglutarate, wo²- Br (22) NO3-, (28) maleate, (29) SO42-,- fumarate, (33) oxaloacetate, (34) WO42, AsO SO² Mo0² (38) As 043- (44)1-I. 40 (26) malonate, (27) citraconitate, (31) C2042-C₂0² * (32) PO³ 1 (36) PO43- * (41) iso-citrate, (42) cis-aconitate, (43) trans-aconitate, > (37) phthalate,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Answer a The probable charge of pyruvate peak 1010 is 2 the probable charge of ...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
Iodide ion is oxidized by hypochlorite ion in basic solution. I(aq) + ClO(aq) Cl(aq) + IO(aq) In 1.00 M NaOH at 25oC, the iodide-ion concentration (equal to the ClO concentration) at different times...
-
Consider the reaction of peroxydisulfate ion (S2O8-2) with iodide ion (I) in aqueous solution: S2O82- (aq) + 3 I (aq) - 2 SO42- (aq) + I3(aq) At a particular temperature the initial rate of...
-
The iodide ion catalyzes the decomposition of aqueous hydrogen peroxide, H2O2. This decomposition is believed to occur in two steps. H2O2 + I H2O + IO (elementary reaction) H2O2 + IO H2O + O2 + I...
-
Clara Hughes, who is pushing 5 0 , has medaled both in speedskating and road cycling ( and showing no signs of slowing down ) completed a training event where she biked 5 0 km east, stopped and rode...
-
For the conditions of Exercise 7 and the data given in Table 11.1, determine the value of R2, as defined by Eq. (11.5.26).
-
What is a post-audit, and what is the purpose of this audit?
-
You are Br erired: with the following information relating to Plott plc for the year to 31 March 2011: Additional information: 1. The above information has been obtained after the compilation of the...
-
Austin Enterprises makes and sells three types of dress shirts. Management is trying to determine the most profitable mix. Sales prices, demand, and use of manufacturing inputs follow: The company...
-
Durai joined in organization for a fortnight and he had conversation with Raj. Durai was hired as company counselor; felt that he was fed up with Raj's team of non-performa. There had been two major...
-
Consider trade relations between the United States and Mexico. Assume that the leaders of the two countries believe the payoffs to alternative trade policies are as follows: a. What is the dominant...
-
Isocratic separation with HPLC Teaching Assistant, an Excel spreadsheet that simulates a reversed-phase liquid chromatographic separation. Download the Excel file from SaplingPlus,...
-
Simulating a separation with a spreadsheet. Use the spreadsheet in Figure 25-39 to simulate the chromatograms for =0.75= 0.75 and =0.56= 0.56 in Figure 25-40. Figure 25-39 Figure 25-40 A 1...
-
Describe a disclosure checklist. What is its purpose?
-
Company Audi's capital structure includes Equity, Preference shares and Bonds (Debt). It wants to calculate its WACC (Weighted average cost of capital) Capital Structure: The market value of Audi's...
-
1. The firm imports high quality wool to France from foreign suppliers located in Australia and the New Zealand. 2. The firm exports leather goods to USA from France. 3. The firm exports designer...
-
In the 8-puzzle game, assume that the start state is: 2 3 4 5 7 6 1 8 An agent is to be designed to solve the puzzle. Answer the following questions: 1. Is the environment fully observable? Explain...
-
Find the derivative of the function. 54 s(t) = 47 (5t-9)
-
What are the key challenges and opportunities associated with fostering diversity, equity, and inclusion within organizational structures, and how can these principles be integrated into talent...
-
The following accounts are denominated in pesos as of December 31, 2013. For reporting purposes, these amounts need to be stated in U.S. dollars. For each balance, indicate the exchange rate that...
-
The executor of Gina Purcells estate has recorded the following information: Assets discovered at death (at fair value): Cash . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
Find the absolute and percent relative uncertainty and express each answer with a reasonable number of significant figures. (a) 9.23 ( 0.03) + 4.21 ( 0.02) - 3.26 ( 0.06) = ? (b) 91.3 ( 1.0) 40.3 (...
-
Verify the following calculations: (a) 3.1415( 0.0011) = 1.77243( 0.00031) (b) log[3.1415 ( 0.0011)] = 0.49714 ( 0.00015) (c) antilog[3.141.5 ( 0.0011)] = 1.3852( 0.0035) 103 (d) ln[3.1415 (...
-
Verify the following calculations: (a) 3.1415( 0.0011) = 1.77243( 0.00031) (b) log[3.1415 ( 0.0011)] = 0.49714 ( 0.00015) (c) antilog[3.141.5 ( 0.0011)] = 1.3852( 0.0035) 103 (d) ln[3.1415 (...
-
A venture's social capital consists of the firm's Blank______. Multiple choice question. external contacts and advisers tangible and intangible assets managers and employees financial resources
-
According to the Fair Labor Standards Act of 1938, overtime provision requires payment at one-and-a-half times the standard for working more than: Multiple choice question. 12 hours per day. 24 hours...
-
All three capital lease criteria must be met in order for a lease to be deemed a capital lease. Group startsTrue or FalseTrue, unselectedFalse, unselectedGroup ends

Study smarter with the SolutionInn App


