Make a graph of [Ag + ], [AgOH(aq)], [CN - ], and [HCN] as a function of
Question:
Make a graph of [Ag+], [AgOH(aq)], [CN-], and [HCN] as a function of pH in a saturated solution of AgCN.
Consider the following equilibria and do not consider activity coefficients. Find the pH if no buffer were added.
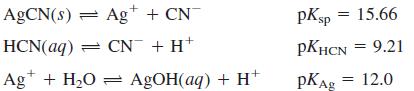
Transcribed Image Text:
AGCN(s) = Ag+ + CN pKsp = 15.66 HCN(aq) = CN + H+ PKHCN = 9.21 Ag* + H2O = AGOH(aq) + H pKAg = 12.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
To create a graph of the concentrations of Ag AgOHaq CN and HCN as a function of pH in a saturated solution of AgCN we need to consider the given equi...View the full answer

Answered By

M Ali Hasan
As a student tutor i am very proud to helping other students and sharing my knowledge to the students community I am very happy to work in solutioninn.com
As a tutor if they provide me the job I m very glad to provide the quality education and quick step by step answers to all the students
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Consider the following equilibria in aqueous solution: (a) Calculate the numerical value of the equilibrium constant for the reaction (b) Calculate the concentration of AgCl(aq) in equilibrium with...
-
Find the concentrations of Ag+(aq), NH3(aq), and [Ag(NH3)2]+(aq) at equilibrium when 0.10 mol Ag+(aq) and 0.10 mol NH3(aq) are made up to 1.00 L of solution. The dissociation constant, Kd, for the...
-
The formation constant for the reaction Ag+ + 2NH3 [Ag(NH3)2]+ is 1.5 Ã 107 and that for the reaction Ag+ + 2CN- [Ag(CN)2]- is 1.0 Ã 1021 at 25°C (see Table 16.3). Calculate the...
-
Using atomic weight, crystal structure, and atomic radius data tabulated inside the front cover of the book, compute the theoretical densities of aluminum (Al), nickel (Ni), magnesium (Mg), and...
-
Independent samples of sizes nA = 2 and nB = 2 are taken from two continuous populations. (a) Enumerate all possible collections of ranks associated with population A. Also attach probabilities to...
-
The following information is available from the financial statements of the Philips Company. Compute the current ratio and the debt-to-total-assets ratio and comment on any trends observed between...
-
Explain personal financial planning and its importance.
-
Installment-Sales Computations and Entries Presented below is summarized information for Johnston Co., which sells merchandise on the installment basis. (a) Compute the realized gross profit for each...
-
. Waterway Co. sells product P-14 at a price of $48 a unit. The per-unit cost data are direct materials $17, direct labour $12, and overhead $16 (75% variable). Waterway has no excess capacity to...
-
A solid block X of mass MX may be placed at different locations along a curved ramp. At the bottom of the ramp is a solid block Y of mass MY that is at rest on a horizontal surface. Figure 1 shows...
-
(a) Find the concentrations of species in saturated CaF 2 as a function of pH by using Reactions 12-32 through 12-36 and adding the following reaction: Do not include activity coefficients. Produce a...
-
Difference plot. A solution containing 3.96 mmol acetic acid plus 0.484 mmol HCI in 200 mL, of 0.10 M KC1 was titrated with 0.490 5 M NaOH to measure K. for acetic acid. (a) Write expressions for the...
-
For each of the service business transactions of a dentist listed below, you are to enter in the Workbook: a the account name, with the debit account first b whether the account entry is a debit or...
-
The following are selected ledger accounts ofMetlockCorporation at December 31, 2017. Cash$188,470 Salaries and wages expense (sales)$287,470 Inventory538,470 Salaries and wages expense...
-
Simplify each expression. 7. -532+718 9.-3-56+189 8. 224-812+754 10. 472x-3x242x
-
A company sells its single product for $40 per unit. The company's after-tax net incomefor the past year was $1,188,000 after applying an effective tax rate of 40%. The projected costs for...
-
SimplengTaksil Company produces furniture. It has four products, tables, chairs, cabinets and bookshelves. It has 50,000 direct labor hours available. Below are the details pertaining to each...
-
What are the key drivers of employee engagement and job satisfaction in contemporary organizations, and how can HR practitioners and managers design and implement effective employee engagement...
-
A political candidate wants to make a mass mailing to counteract some nasty remarks made recently by her opponent. Our candidate has identified five mailing lists that contain names and addresses of...
-
San Carlos Bank and Trust Company uses a credit-scoring system to evaluate most consumer loans that amount to more than $2,500. The key factors used in its scoring system are found at the conclusion...
-
What is selected reaction monitoring? Why is it also called MS/MS? Why does it improve the signal/noise ratio for a particular analyte?
-
(a) To detect the drug ibuprofen by liquid chromatography/ mass spectrometry, would you choose the positive or negative ion mode for the spectrometer? Would you choose acidic or neutral...
-
An electrospray/transmission quadrupole mass spectrum of the -chain of hemoglobin from acidic solution exhibits nine peaks corresponding to M n+ n . Find the charge, n, for peaks A-I. Calculate the...
-
Several points are shown on the complex plane. Imaginary Axis P 8 7 6 5 4 21 3 2 1 -14-13-12-11-10 -9 -8 -7 -6-5-4 -3 -2 S R Which point represents Z + Z? OP OQ OR Os Real axis -1 1 2 3 4 5 -1 -2 -3...
-
Determine the domain of (g. f)(x) if f (x) = x + 3x5 and g(x)= OXER) xERX-4,-1,1) (xRx-4, 1) (xRxz-1) (3 points) X+1
-
Exercise: Journal Practice 1 Exercise: Journal Practice 1 - - -- Directions The following is a list of financial transactions that occurred in your medical practice during the month of January. 1....

Study smarter with the SolutionInn App


