On the basis of the following reaction, state which compound, H 2 (g) or glucose, is the
Question:
On the basis of the following reaction, state which compound, H2(g) or glucose, is the more powerful reducing agent at pH = 0.00.
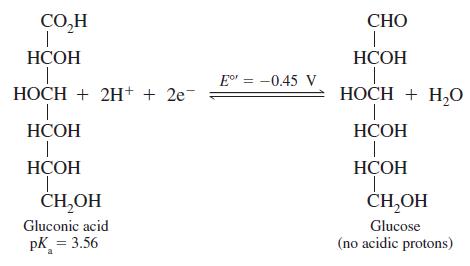
Transcribed Image Text:
CO,H СНО НСОН НСОН Eo = -0.45 V НОСН + 2H+ + 2e- НОСН + H,О НСОН НСОН НСОН НСОН 1. ČH,OH CH,OH Gluconic acid Glucose pK = 3.56 (по аcidic protons)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
In this reaction Gluconic acid reduced into glucose So we can say that Gluconic acid ...View the full answer

Answered By

Vijay Kumar
I work as a tutor , I explain answer of any question as simple language and students understand thats my answer and explanation. I use simple wording when give any answer to students....I love teaching..
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Complete the following reaction. Decide on the basis of relative acid strengths whether the reaction is more likely to go in the direction written or in the opposite direction. HS + HCN
-
Which compound will undergo an electrophilic aromatic substitution reaction more rapidly, benzene or hexadeuteriobenzene? or D- H.
-
Given the following data: Calculate ÎH for the reaction On the basis of enthalpy change, is this a useful reaction for the synthesis of ammonia? AH - 92 kJ () + AH = -484 k (g) ON OH 88
-
What are the pros and cons of using social media at work? Discuss.
-
A shipment of assorted nuts is labeled as having 45% walnuts, 20% hazelnuts, 20% almonds, and 15% pistachios. By randomly picking several scoops of nuts from this shipment, an inspector finds the...
-
Chipman Corporation expects to incur \($300,000\) of factory over-head and \($500,000\) of general and administrative costs next year. Direct labor costs at \($25\) per hour are expected to total...
-
In this exercise, we explore what happens when an item is marked up by a percentage, and then marked down using the same percentage. Wegmans purchases an item for \(\$ 5\) per unit. The markup on the...
-
Olive Corporation was formed and began operations on January 1, 2014. The corporations income statement for the year and the balance sheet at year-end are presented below. The corporation made...
-
3. In some cases, preferred stock has some characteristics of a debt instrument. a. How should the accountant classify such debt in the financial instrument? b. Reference relevant authorities from...
-
Hughs Repair Shop specializes in repairing German and Japanese cars. The shop has two mechanics. One mechanic works ononly German cars and the other mechanic works on only Japanese cars. In either...
-
The formation constant for Cu(EDTA) 2+ is 6.3 10 18 , and E is +0.339 V for the reaction From this information, find E for the reaction Cu+ + 2e Cu(s).
-
Why might an organization seek the assurance of a CPA about information presented in the organizations corporate sustainability report?
-
When the financial controller of Bakers Company set the budget for the year ahead, it was expected that monthly output of cake packages would be 12,000 units. In March the output was increased to...
-
The fixed and variable costs for three potential manufacturing plant sites for a rattan chair weaver are shown: a) After rounding to the nearest whole number, site 1 is best below After rounding to...
-
Classic Cabinets has a factory that produces custom kitchen cabinets. It has multiple product lines. Materials and labor for the cabinets are determined by each job. To simplify the assignment, we...
-
Sheffield power produces and sells customs lawn mowers for $202 and has a contribution margin of $123 per unit. monthly fixed cost are $32320 the cost equation for sheffield power lawn mowers is
-
Please answer Suppose consumers' disposable income increased by $500 billion and their spending increased by $430 billion. What was the MPC? Instructions: Round your response to two decimal places....
-
On april 1 , 2 0 2 0 , Nair manufacturing purchased a machine for $ 5 0 0 , 0 0 0 and made the following estimates: a ) residual value $ 8 0 , 0 0 0 b ) service life 5 years c ) estimated...
-
In 2017, Chen Corporation purchased treasury stock with a cost of $46,000. During the year, the company declared and paid dividends of $14,000 and issued bonds payable for $900,000. Net cash provided...
-
You are the newly appointed tax practitioner to complete Emilys tax return and have downloaded the prefill report for Emilys tax return (hint, you can read what a prefill report is here (Links to an...
-
Morphine and morphine 3-β-D-glucuronide were separated on two different 4.6-mm diameter à 50-mm-long columns with 3-μm particles.39 Column A was C18-silica run at 1.4...
-
The rate at which heat is generated inside a chromatography column from friction of flowing liquid is power (watts, W = J/s) = volume flow rate (m3/s) pressure drop (pascals, Pa = kg/[m s2]). (a)...
-
Cocaine metabolism in rats can be studied by injecting the drug and periodically withdrawing blood to measure levels of metabolites by HPLC-mass spectrometry. For quantitative analysis, isotopically...
-
The correct formula of the compound whose name is hexaamminechromium(III) nitrate is a.[Cr(NO3)](NH3)6 b.[Cr(NH3)6](NO3) c.[Cr(NO3)3](NH3)6 d.[Cr(NH3)6(NO3)3] e.[Cr(NH3)6](NO3)3
-
1. Data-driven decision making tends to decrease a firm's a. market value. b. productivity. c. risk. d. profit.
-
The ratio of a persons body weight to height is represented as his or her a. body composition. b. basal metabolic rate. c. bioelectrical impedance. d. body mass index. 2: Energy balance occurs when...

Study smarter with the SolutionInn App


