Using activities, find the concentrations of the major species in 0.10 M NaClO 4 saturated with Mn(OH)
Question:
Using activities, find the concentrations of the major species in 0.10 M NaClO4 saturated with Mn(OH)2. Take the ionic strength to be 0.10 M and suppose that the ion size of MnOH+ is the same as Mn2+. Consider just the following chemistry:
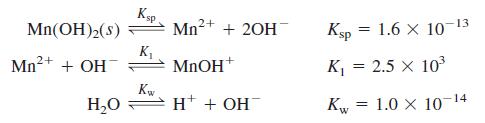
Transcribed Image Text:
Ksp Mn2+ 13 Mn(OH)2(s) + 20H Ksp = 1.6 X 10 Mn2+ + OH K, MNOH* K = 2.5 x 103 К H* + OH 1.0 x 10- Kw -14 H,0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
Or H Mn OHS Ksp Mn 2OH Or H Ksp 161013 Mn OHK Mn ...View the full answer

Answered By

Saurav Solanki
I love physics . After i did btech(grad) in electronics , started teaching physics as a home tutor , later in 2017 i satrted tutoring online. I have taught in Chegg , 24houranswers.com . Learning physics concept is easy when taught with practical examples . With my 3 yrs of experience , i want you to give me chance to teach you in a better way.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Use the systematic treatment of equilibrium to find the concentrations of the major species in a saturated aqueous solution of LiF. Consider these reactions: LiF(s) = Li* +F Ksp = [Li*]YLi+[F]YF Kjon...
-
Find the concentrations of Ag+(aq), NH3(aq), and [Ag(NH3)2]+(aq) at equilibrium when 0.10 mol Ag+(aq) and 0.10 mol NH3(aq) are made up to 1.00 L of solution. The dissociation constant, Kd, for the...
-
(a) Find the concentrations of species in saturated CaF 2 as a function of pH by using Reactions 12-32 through 12-36 and adding the following reaction: Do not include activity coefficients. Produce a...
-
At its headquarters in Ventura, California, Patagonia's office space feels more like a national park lodge than the main office of a $400 million retailer. It has a Douglas fir staircase and a...
-
A developer acquired a parcel of unimproved real property that she would like to develop. Although the land is currently zoned for commercial use, the developer would prefer not to begin development...
-
Equipment costing \($290,000,\) with an expected scrap value of \($30,000\) and an estimated useful life of five years, was purchased on January 1, 2016. Calculate the depreciation expense for years...
-
Compare the schemes introduced for the linear convection equation. a) Simple explicit schemes (7.6), (7.7), and (7.8) b) Simple implicit scheme (7.19) c) Leapfrog scheme (7.21) d) Lax-Wendroff scheme...
-
The Britts & Straggon company manufactures small engines at three different plants. From the plants, the engines are transported to two different warehouse facilities before being distributed to...
-
Prepare an amortization schedule for a five-year loan of $42,000. The interest rate is 8% per year, and the loan calls for equal annual payments. (Do not round intermediate calculations. Enter all...
-
Consider the composite solid shown. Solid A is a thermally conductive material that is 0.5-cm thick and has a thermal conductivity, k A = 50 W/m K. The back side of solid A (x = 0) is thermally...
-
Write charge and mass balances for aqueous Ca 3 (PO 4 ) 2 if the species are Ca 2+ , CaOH + , CaPO - 4 , PO 3- 4 , HPO 2 4 - , H 2 PO - 4 , and H 3 PO 4 .
-
Explain why the solubility of an ionic compound increases as the ionic strength of the solution increases (at least up to ~ 0.5 M).
-
Bires Ronsard SA recently purchased a brewing plant from a bankrupt company. The brewery is in Montpazier, France. It was constructed only two years ago. The plant has budgeted fixed manufacturing...
-
Jackson Co. has the following information for the first 4 months of this year: Machine hours Cleaning Hours Expense January 2,100. $900 February 2,600 1,200 March 1,600 800 April 2,000 1,000...
-
Identify the problem statement for Apple bites back. Next, identify what will Apple need to do to maintain product innovation and customer loyalty? How has Apple developed extreme loyalty among...
-
X Company currently makes 2,600 units of a unique part for one of its finished products. Variable production costs for this part are $12.12 per unit; fixed costs associated with this part are $16,000...
-
what is Risk Classification of data (based on sensitivity of data and likelihood for malicious purposes) - CRD (Critical Risk data), HRD (High Risk Data), and MRD (Medium Risk Data) In Uber business...
-
Write a letter to the managing director of a company explaining relevant costing and any problems of this approach.
-
1. Prepare a budget for your project. A budget should be very inclusive of all cost the project plans to incur throughout the duration of the project including a contingency allowance. 2. Prepare a...
-
d. The characteristic equation of a control system is given by s+2s+8s+12s+20s+16+16=0. Determine the number of the roots of the equation which lie on the imaginary axis of s-plane
-
Calculate pCo2+ at each of the following points in the titration of 25.00 mL of 0.020 26 M Co2+ by 0.03855 M EDTA at pH 6.00: (a) 12.00 mL; (b) Ve; (c) 14.00 mL.
-
Consider the titration of 25.0 mL of 0.0200 M MnSO4 with 0.010 0 M EDTA in a solution buffered to pH 8.00. Calculate pMn2+ at the following volumes of added EDTA and sketch the titration curve: (a) 0...
-
For the same volumes used in Problem 11-8, calculate pCa2+ for the titration of 25.00 mL of 0.02000 M EDTA with 0.010 00 M CaSO4 at pH 10.00.
-
negg G what is x | G budge x How T. X Sign ox Refere x S Week X G Whic ontent/107694/viewContent/5524273/View When there is a reserve requirement, banks a) must hold exactly the required quantity of...
-
Lily is a college student who likes to buy only two goods: Cheetos and Pepsi. To determine Lily's budget line, you need to know I. Lily's preferences for Cheetos and Pepsi. II . The prices of Cheetos...
-
Chapter 3- Labor Productivity and Comparative - Advantage: The Ricardian Model 1. Home has 1,200 units of labor available. It can produce two goods, apples and bananas. The unit labor requirement in...

Study smarter with the SolutionInn App


