What is the purpose of the WO3 WO 3 and Cu Cu in Figure 27-8? Figure 27-8
Question:
What is the purpose of the WO3WO3 and CuCu in Figure 27-8?
Figure 27-8
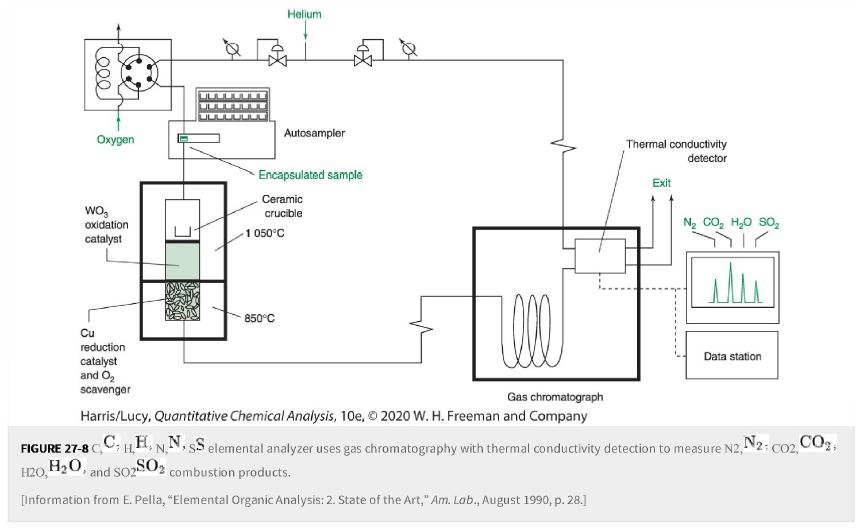
Transcribed Image Text:
Leee Oxygen WO oxidation catalyst Cu reduction catalyst and O₂ scavenger 7. Encapsulated sample Ceramic crucible Helium Autosampler 1 050°C 850°C Well Gas chromatograph Harris/Lucy, Quantitative Chemical Analysis, 10e, © 2020 W. H. Freeman and Company Thermal conductivity detector FIGURE 27-8 C H20, H₂O, and S02 SO2 combustion products. [Information from E. Pella, "Elemental Organic Analysis: 2. State of the Art," Am. Lab., August 1990, p. 28.] Exit Ng CO, HO SO, Sair Data station CHEN. HH, NN, SS elemental analyzer uses gas chromatography with thermal conductivity detection to measure N2, N2: CO2, CO₂,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
The WO3WO3 and CuCu combination is placed in a single process in ord...View the full answer

Answered By

Tamondong Riza
Professionally, I am a teacher with years of experience tutoring math and science, as well as teaching in both public schools and independent schools. I feel that education should be an enlightening experience for all children, and I'm committed to helping my students learn new skills and make progress in their subjects.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
What is the purpose of IFRS 8 on Operating Segments?
-
What is the purpose of application controls? How does it differ from the purpose of general controls?
-
What are transportation user charges? What is the purpose of such charges?
-
Elmira, Inc had $20,000,000 of bonds outstanding on Dec 31, 2018. The ten year bonds were issued on Jan 1, 2012 for $21,000,000. Elmira can call the bonds at 102% any time after Jan 1, 2018. At the...
-
Consider a multiple linear regression problem with design matrix Z and observations Y. Let Z1 be the matrix remaining when at least one column is removed from Z. Then Z1 is the design matrix for a...
-
Discuss how companies manage their international marketing activities.
-
Identify methods to resolve consumer complaints.
-
On June 30, 2009, Mischa Auer Company issued $4,000,000 face value of 13%, 20-year bonds at $4,300,920, a yield of 12%. Auer uses the effective interest method to amortize bond premium or discount....
-
Choose the letter of the correct answer Case 2 You have been engaged to review the records Excalibur Corporation, a small manufacturer and precision tools and machines. Your examination reveals the...
-
The accounts below all have normal balances. Service Fees $40,650 Accounts Receivable 18,000 Rent Expense 3,700 Utilities Expense 1,400 Alana, Capital 42,100 Alana, Drawing 2,700 Wages Expense 18,300...
-
Write a balanced equation for combustion of C 8 H 7 NO 2 SBrCl C 8 H 7 NO 2 SBrCl in a C,C,H,H,N,N,SS elemental analyzer.
-
Corrected peak area. A micellar electrokinetic chromatography method for phthalazine and its metabolite 1-1- phthalazinone yielded the following repeatability for six replicate injections of a 50 M...
-
Given the following information, determine the equivalent units of ending work in process for materials and conversion under the weighted-average method: Beginning inventory of 2,500 units is 100%...
-
Sunn Company manufactures a single product that sells for $220 per unit and whose variable costs are $176 per unit. The company's annual fixed costs are $664,400. (1) Prepare a contribution margin...
-
A design team is in the process of designing a gyro-stabilizer for a boat against rolling. Motor 4 in Figure I takes the signal from the water wave and turns the large precession gear B up to a...
-
What are the pivotal factors influencing the successful implementation of transformative initiatives within complex organizational structures ? Explain
-
Recently, a meme on social media was divvying up lottery winnings among everyone in the country. If the jackpot at that time was $2.2 billion and the meme estimated a population of 300 million...
-
Who Am I? The Who Am I exercise is designed to get students to think about the worldview and perspective of other cultures based on their histories. This is a role-playing exercise. Please choose one...
-
In government-wide financial statements, how do intra-activity and interactivity transactions differ? How is each type of transaction reported?
-
Ball bearings are widely used in industrial applications. You work for an industrial food machinery manufacturer and your role is to design the driveshaft assembly on a new type of equipment that...
-
(a) Use the ideal gas law (Problem 1-16) to calculate the density (g/mL) of helium at 20C and 1.00 bar. (b) Find the true mass of Na (density = 0.97 g/mL) weighed in a glove box with a He atmosphere,...
-
(a) The equilibrium vapor pressure of water at 20C is 2330 Pa. What is the vapor pressure of water in the air at 20C if the relative humidity is 42%? (Relative humidity is the percentage of the...
-
Effect of altitude on electronic balance. If an object weighs ma grams at distance ra from the center of the Earth, it will weigh mb = ma(r2a/r2b) when raised to rb. An object weighs 100.0000 g on...
-
What types of information can be found in a mutual fund prospectus? Multiple select question. information about dividends and distributions information about current net asset value information about...
-
Instructions Using the information from previous chapters and the new information above, do the following: a. Answer Natalie's questions. b. Prepare and post to T accounts the June 2024 transactions....
-
Concerning factoring, all of the following are true except: Group of answer choices factors prefer business over consumer accounts factoring is done at a discount to the third party purchaser...

Study smarter with the SolutionInn App


