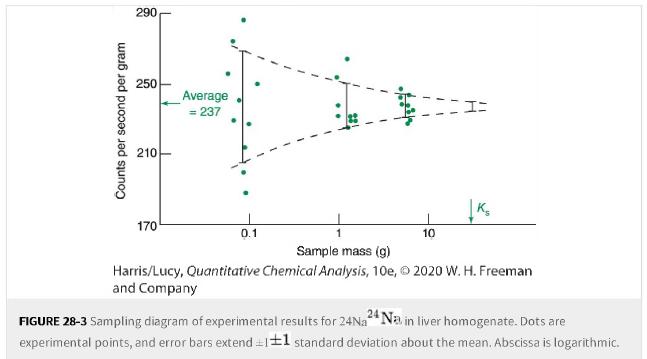
What mass of sample in Figure 28-3 is expected to give a sampling standard deviation of 6%?6%?
Question:
What mass of sample in Figure 28-3 is expected to give a sampling standard deviation of ±6%?±6%?
Figure 28-3
Transcribed Image Text:
Counts per second per gram 290 250 210 170 Average = 237 0.1 10 K₂ Sample mass (g) Harris/Lucy, Quantitative Chemical Analysis, 10e, 2020 W. H. Freeman and Company FIGURE 28-3 Sampling diagram of experimental results for 24Na 24 Na in liver homogenate. Dots are experimental points, and error bars extend = 1±1 standard deviation about the mean. Abscissa is logarithmic.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (16 reviews)
Calculations The mass of sample in Figure 283 is expected to give ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
Louvre Inc. bought a business that is expected to give a 25% annual rate of return on the investment. Of the total amount paid for the business, $75,000 was deemed to be goodwill, and the rest was...
-
Which of the following would be expected to give a positive test with Benedict's reagent? Why? (a) L-Arabinose (b) 1,3-Dihydroxyacetone (c) D-Fructose (d) Lactose (e) Amylose
-
What are the mean and standard deviation of the sampling distribution of x ? What are the mean and standard deviation of the sampling distribution of p?
-
1. A projectile is launched in a vertical plane, at an angle 0 with initial velocity vo. It must be caught in a frictionless circular tube of radius R in such a way that the trajectory of the...
-
Suppose that X1, . . . , Xm form a random sample from a continuous Distribution for which the p.d.f. f (x) is unknown; Y1, . . . , Yn form an independent random sample from another continuous...
-
Describe alternative ways to prorate end-of-period adjustments for under-or over allocated indirect costs.
-
Go to the Securities and Exchange Commission's Internet site (sec.gov) and find a Litigation Release number that deals with each of the following issues. Give the Litigation Release number and write...
-
Carlton Company is involved in four separate industries. The following information is available for each of the four industries. Instructions Determine which of the operating segments are reportable...
-
A. Each purchase order specifies many products O B. Each purchase is for one product C. Checks are written after products are added to inventory D. Each purchase can include many products QUESTION 11...
-
Suppose you are gambling on a roulette wheel. Each time the wheel is spun, the result is one of the outcomes 0, 1, and so on through 36. Of these outcomes, 16 are red, 16 are black, and 1 is green....
-
Cloud-point extraction. Traces of lead in drinking water cause adverse health effects. The micelle-forming surfactant Triton X-114 (plus the co-surfactant cetyl trimethylammonium bromide) and a crown...
-
In the chapter opener, trace metals in teeth and bone provide information about Otzi the Icemans diet and environment as a child and adult. The inorganic mineral matrix of teeth and bone is...
-
Suppose the function F(x, t) satisfies with terminal condition: F(X(T),T) = h(X(T)). Show that F(x, t) = e r (T t) E t [h(X(T))|X(t) = x], t a F - at a F + m(x, t). x + o(x, t) 0 F 2 2 r F = ()
-
You borrowed 20,000 from a local credit union to buy a car. You promised to pay this loan in 5 years. Suppose 3 years lato your loan you have decided to sell the 10, 000 When you sell the car, you...
-
Describe to the client why workforce planning is the first key step to the audit, and beneficial to their company. Identify and describe the relevant job and job classification, job family. Identify...
-
How does a changing interest rate affect the value of money over time? In the Ginny's restaurant case, how do we leverage potential future cash flows to attain funds today? How would our decisions...
-
Brazil is very proud of its aircraft manufacturing capabilities, particularly with the company, Embraer, which specializes in passenger aircraft holding between _____ passengers. Group of answer...
-
9. Assume the normal PDF is a good model for stock returns. Your firm has created a "fund" (a portfolio of stocks) with an expected annual return of 13% and a standard deviation of 0.18. Risk-free...
-
Akron, Inc., owns all outstanding stock of Toledo Corporation. Amortization expense of $15,000 per year for patented technology resulted from the original acquisition. For 2013, the companies had the...
-
In July 2013, cnet.com listed the battery life (in hours) and luminous intensity (i. e., screen brightness, in cd/m2) for a sample of tablet computers. We want to know if screen brightness is...
-
Calculate how many milliliters of 0.626 M KOH should be added to 5.00 g of MOBS (Table 8-2) to give a pH of 7.40.
-
(a) Use Equations 8-20 and 8-21 to find the pH and concentrations of HA and A- in a solution prepared by mixing 0.00200 mol of acetic acid plus 0.004 00 mol of sodium acetate in 1.00 L of water. (b)...
-
(a) Calculate the pH of a solution prepared by mixing 0.0100 mol of the base B (Kb = 10 = - 2.00) with 0.020 0 mol of BH+Br- and diluting to 1.00 L. First calculate the pH by assuming [B] = 0.0100...
-
Which is true of adding a video to a Google Slides presentation? O you need to have the URL in order to add a video directly, otherwise, you have to download it to your computer first O you can...
-
Which statement about competency models is true? OThey help HR professionals ensure that all aspects of talent management are aligned with an organization's strategy. OThey identify and describe a...
-
Good time management includes always completing your easiest task first which will create a sense of accomplishment and motivate you to tackle next the most difficult task. True False

Study smarter with the SolutionInn App


