What is the coefficient of O2 ? 5 O 10 11 O 4 FeS2 + 02...
Fantastic news! We've Found the answer you've been seeking!
Question:
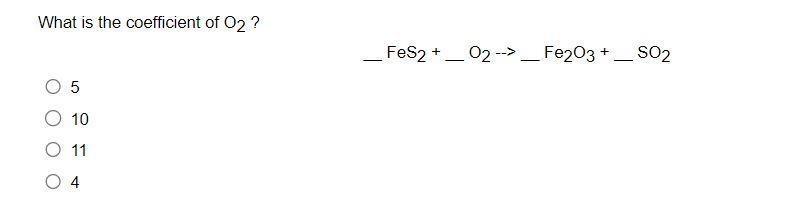

Transcribed Image Text:
What is the coefficient of O2 ? 5 O 10 11 O 4 FeS2 + 02 --> - Fe2O3 + SO2 - For a system that gains thermal energy and does work on the surroundings, heat (q) is negative and work (w) is positive. heat (q) is positive and work (w) is negative. heat (q) and work (w) are both negative. heat (q) and work (w) are both positive. What is the coefficient of O2 ? 5 O 10 11 O 4 FeS2 + 02 --> - Fe2O3 + SO2 - For a system that gains thermal energy and does work on the surroundings, heat (q) is negative and work (w) is positive. heat (q) is positive and work (w) is negative. heat (q) and work (w) are both negative. heat (q) and work (w) are both positive.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Mary plans to sell two different software products: X and Y. Assume the budgeted sales mix (3 units of X sold for every 2 units of Y sold) will not change. A total of 100 units (60 units from product...
-
Write a C++ singly linked list program which asks the user to provide the following information for as many airports as they like: 1). Standard 3 character airport code. 2). Distance from New York...
-
Select any Company and get a copy of its annual report. Just write an outline of the company's annual report. The outline must contain the following: 1. Introduction 2. Background 3. Liquidity Ratios...
-
The following sample data have been collected from two independent samples from two populations. Test the claim that the second population median will exceed the median of the first population. a....
-
Construct three-period and four-period centered moving averages for the quarterly J. C. Penney sales in Exercise 18.67. Are there any other retailing companies for whom a four-period moving average...
-
CaTiO 3 , a perovskite, has the structure below. (a) If the density of the solid is 4.10 g/cm 3 , what is the length of a side of the unit cell? (b) Calculate the radius of the Ti 4+ ion in the...
-
Which would not be a preventive control? a. Guards b. Swipe cards c. Fences d. Data mining software e. Background checks
-
The cash register tape for Bluestem Industries reported sales of $6,871.50. Record the journal entry that would be necessary for each of the following situations. (a) Cash to be accounted for exceeds...
-
Sunland Spoon Service repairs commercial food preparation equipment. The following budgeted cost data is available for 2022: Time Charges Material Charges Technicians' wages and benefits $580,000...
-
Baer Belly Bikinis, Inc. (BBB) is a small business located in Santa Monica, California. It sells swimwear and related products to specialty stores throughout the United States. It also sells its...
-
Canfield, Inc. Comparative Balance Sheet December 31, 2018 and 2017 2018 Assets Current Assets: Cash Accounts Receivables, Net Merchandise Inventory Total Assets Prepaid Expenses Total Current Assets...
-
Assume a company has 5 million shares of common stock outstanding and intends to issue another 1,000,000 shares through a rights offering. The rights will carry a subscription price of $80. If the...
-
Find an upper bound for [Es in estimating O 5 (8x5-2x) dx with n = 10 steps.
-
In the fifth grade at Lenape Elementary School, there are 41 as many girls as there are boys. 7 There are 66 students in the fifth grade. How many students are girls? >> Solve on paper. Then check...
-
For each of the following, compute the present value. (Do not round intermediate calculations and round your answers to 2 decimal places, e.g., 32.16.) Present Value Years Interest Rate Future value...
-
y = (5+ cos(x))* Use Logarithmic Differentiation to find dy dx dy dx
-
Please help me soon! Note: Please save these 2 Python file as a PDF and take a screenshot of that PDF. Also, copy the code and upload it in your answer. Thanks so much! 1. Please write the code tha...
-
B.) What is the approximate concentration of free Zn 2+ ion at equilibrium when 1.0010 -2 mol zinc nitrate is added to 1.00 L of a solution that is 1.080 M in OH - . For [Zn(OH) 4 ] 2- , K f = 4.610...
-
The 12 smartphones in Table 26 have been randomly selected for the sample of an experiment. Use the seed 994 to randomly select 6 of the phones for the treatment group. The other 6 phones will be in...
-
The simulations of flipping a coin 5 times and an additional 10,000 times are shown in Figs. 9 and 10, respectively. a. Determine the first five outcomes of the simulated experiment. b. What was the...
-
Simvastatin is a prescription drug used to lower blood cholesterol. However, researchers tested whether the drug also heals ulcers. A total of 66 ulcer patients were tested. The study concluded that...
-
You are to draw up a consolidated balance sheet as at 31 December 2005 from the following: At the balance sheet date, Son 1 owed Pa and Mum 2,500 and Son 2 1,100, and Pa and Mum owed Son 2 2,100. Pa...
-
You are presented with the following information from the Seneley group of companies for the year to 30 September 2006: Additional information: (a) The authorised, issued and fully paid share capital...
-
Prepare a consolidated balance sheet from the following details as at 31 March 2003. At the balance sheet date, Filho B owed Filho A 3,000 and Pops for Filhos owed Filho B 2,000. During the year,...

Study smarter with the SolutionInn App



