A 100 kmol of natural gas is burned with 20% of excess pure O. The natural...
Fantastic news! We've Found the answer you've been seeking!
Question:
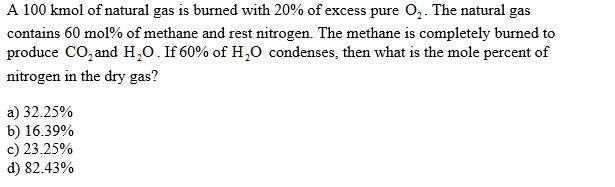
Transcribed Image Text:
A 100 kmol of natural gas is burned with 20% of excess pure O₂. The natural gas contains 60 mol% of methane and rest nitrogen. The methane is completely burned to produce CO₂ and H₂O. If 60% of H₂O condenses, then what is the mole percent of nitrogen in the dry gas? a) 32.25% b) 16.39% c) 23.25% d) 82.43% A 100 kmol of natural gas is burned with 20% of excess pure O₂. The natural gas contains 60 mol% of methane and rest nitrogen. The methane is completely burned to produce CO₂ and H₂O. If 60% of H₂O condenses, then what is the mole percent of nitrogen in the dry gas? a) 32.25% b) 16.39% c) 23.25% d) 82.43%
Expert Answer:
Answer rating: 100% (QA)
Answer Solution sine CH4 20 Metham reaction combustion reaction cot 2420 Took... View the full answer

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these finance questions
-
Natural gas is burned with air to produce gaseous products at 1985C. Express this temperature in K, R and F.
-
Ethane (C 2 H 6 ) is burned with 20 percent excess air during a combustion process. Assuming complete combustion and a total pressure of 100 kPa, determine (a) The airfuel ratio (b) The dew-point...
-
A sample of natural gas is 85.2% methane, CH4, and 14.8% ethane, C2H6, by mass. What is density of this mixture at 18oC and 771 mmHg?
-
(CMA, adapted) Roletter Company makes and sells artistic frames for pictures of weddings, graduations, and other special events. Bob Anderson, the controller, is responsible for preparing Roletters...
-
Lecimore Company has a centralized purchasing department that is managed by Joan Jones. Jones has established policies and procedures to guide the clerical staff and purchasing agents in the...
-
Items 1 through 6 are questions typically found in a standard internal control questionnaire used by auditors to obtain an understanding of internal control for notes payable. In using the...
-
When Escherichia coli (E. coli) is in a broth culture at \(37^{\circ} \mathrm{C}\), the population of \(E\). coli doubles in number with 30 organisms, how many \(E\). coli bacteria are present in the...
-
Conwell Company manufactures its product, Vitadrink, through two manufacturing processes: Mixing and Packaging. All materials are entered at the beginning of each process. On October 1, 2012,...
-
2. Stock Dividends The owners' equity accounts for Hexagon International are shown here: Common stock ($1 par value) Capital surplus Retained earnings Total owners' equity $ 30,000 185,000 627,500...
-
The mean per capita daily water consumption in a village in Bangladesh is about 83 liters per person and the standard deviation is about 11.9 liters per person. Random samples of size 50 are drawn...
-
what were behind the sad and beautiful stories of these establishments? when one fails in business, what was missing? what went wrong? when one succeeds, what are the trade secrets?
-
Explain why L-systems are of interest to Artificial Life researchers.
-
Explain how genetic programming could be used to solve problems.
-
Explain how a system might be built that could reproduce itself. Would such a system be alive?
-
Explain how Conways Life is modeled on life. What interesting properties does it exhibit? Why do you think it has fascinated people for so long?
-
Explain the idea behind meansends analysis. Compare it with search as a planning method. Which more closely matches the methods people use when formulating plans in everyday life?
-
Required information Problem 1 4 - 4 5 Special Order; Financial and Production Considerations ( LO 1 4 - 4 , 1 4 - 5 ) Skip to question [ The following information applies to the questions displayed...
-
Which, if any, of the dichloroethene molecules drawn in Data Table II (3.) (4.) and (5.) are geometric isomers? A. B. C. D. cis-1,2-dichloroethene and trans-1,2-dichloroethene...
-
Why does a nonquasi-equilibrium compression process require a larger work input than the corresponding quasi-equilibrium one?
-
A solar cell has an open circuit voltage value of 0.60 V with a reverse saturation current density of Jo = 3.9 109 A/m2. The temperature of the cell is 27oC, the cell voltage is 0.52 V, and the cell...
-
An adiabatic heat exchanger is to cool ethylene glycol (cp = 2.56 kJ/kg C) flowing at a rate of 2 kg/s from 80 to 40C by water (cp = 4.18 kJ/kg C) that enters at 20C and leaves at 55C. Determine...
-
Consider the two-dimensional Poisson equation (8.6) in a rectangular domain \(0 \leq x \leq L_{x}, 0 \leq y \leq L_{y}\). The boundary conditions are \[\frac{\partial u}{\partial x}(0, y)=g_{1}(y),...
-
Develop a finite difference scheme for the PDE in problem
-
Write the formulas for the Gauss-Seidel algorithm (similar to (8.57)) for the central difference scheme applied to the three-dimensional Poisson equation (8.14).

Study smarter with the SolutionInn App


