A hydrocarbon (containing only carbon and hydrogen) with an unknown formula is subjected to combustion analysis....
Fantastic news! We've Found the answer you've been seeking!
Question:
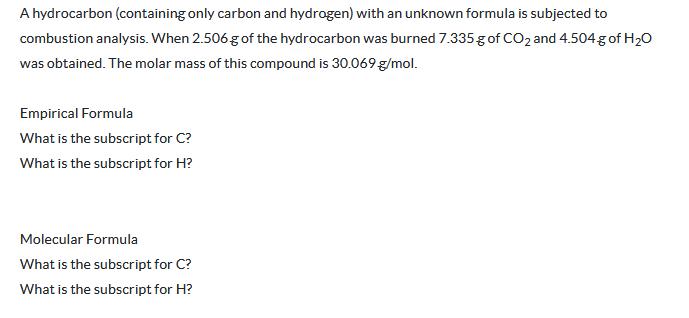
Transcribed Image Text:
A hydrocarbon (containing only carbon and hydrogen) with an unknown formula is subjected to combustion analysis. When 2.506 g of the hydrocarbon was burned 7.335 g of CO2 and 4.504 g of H₂O was obtained. The molar mass of this compound is 30.069 g/mol. Empirical Formula What is the subscript for C? What is the subscript for H? Molecular Formula What is the subscript for C? What is the subscript for H? A hydrocarbon (containing only carbon and hydrogen) with an unknown formula is subjected to combustion analysis. When 2.506 g of the hydrocarbon was burned 7.335 g of CO2 and 4.504 g of H₂O was obtained. The molar mass of this compound is 30.069 g/mol. Empirical Formula What is the subscript for C? What is the subscript for H? Molecular Formula What is the subscript for C? What is the subscript for H?
Expert Answer:
Answer rating: 100% (QA)
To find the empirical formula of the hydrocarbon we first need to determine the moles of carbon and ... View the full answer

Related Book For 

Vector Mechanics for Engineers Statics and Dynamics
ISBN: 978-0073212227
8th Edition
Authors: Ferdinand Beer, E. Russell Johnston, Jr., Elliot Eisenberg, William Clausen, David Mazurek, Phillip Cornwell
Posted Date:
Students also viewed these mechanical engineering questions
-
Draw temperature curves dry air parcels that we release with temperatures of 22C and 24C at the ground. The y-curve in the graph describes the environmental temperature profile. At what altitudes do...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The ABC Company has a cost of equity of 24.76 percent, a before-tax cost of debt of 5.37 percent, and a tax rate of 26 percent. What is the firm's weighted average cost of capital if the proportion...
-
A bag contains 200 balls that are either red or blue and either dull or shiny. There are 55 shiny red balls, 91 shiny balls and 79 red balls. If a ball is chosen at random, what is the probability...
-
10. The following account balances from the adjusted trial balance columns of FABM Trading worksheet are provided below: Account Cash Merchandise Inventory Accounts Payable FAB Mars, Drawing FAB...
-
a. Create a spreadsheet to reconcile bank statements. Organize it as follows: i. Ending balance, per bank statement ii. Deposits not included in bank statement (provide space for up to 7) iii. Checks...
-
This information is available for Paik??s Photo Corporation for 2010, 2011, and 2012.InstructionsCalculate inventory turnover, days in inventory, and gross profi t rate (from Chapter 5) for Paik??s...
-
You have $401492 in a your 401(k) retirement account earning 5.6 percent per year. You are retiring now and have a life expectancy of 8 years. You plan to make equal yearly withdrawals from your...
-
The function (AB) is to be realized using only 2-input NAND gates. The minimum number of 2-input NAND gates required for such a realization is
-
The Phoenix ( P ) Corp. is operating in an essential business that its local council is sponsoring. To induce P Corp to remain in this essential business, the council has agreed to pay whatever...
-
Here is some hypothetical testimony from prosecution of an assault of a victim in a grocery store parking lot: The prosecutor calls Bart (an eyewitness and the "primary" or "fact" witness in this...
-
After the tangible assets have been adjusted to current market prices, the capital accounts of Elayne Summers and Murv Newcomb have balances of $86,000 and $146,000, respectively. Rose Clayton is to...
-
A 100 gram sample of unknown radioactive element decays into non-radioactive substances. In 440 days the radioactivity of a sample decreases so only 63 grams remain. Use the equation, A(t)=Aoekt....
-
what ways has the incorporation of metal-organic frameworks (MOFs) in catalytic applications expanded the scope of catalysis, particularly in gas-phase reactions and environmental catalysis ?
-
In a criminal trial, the prosecution seeks to prove that the defendant, Calvin Broadus robbed a store. There was a witness named Christopher Wallace who told a police officer named Jermaine Cole that...
-
"Wilson Machine Tools is considering a high - tech computer - controlled milling machine at a cost of $ 8 2 , 0 0 0 . The cost of installing the machine, preparing the site, wiring, and rearranging...
-
What impact has the Internet had on the globalization of small firms? How do you think small companies will use the Internet for business in the future?
-
A 2-kg sphere moving to the right with a velocity of 5 m/s strikes at A the surface of a 9-kg quarter cylinder which is initially at rest and in contact with a spring of constant 20 kN/m. The spring...
-
A 1.2-lb ball A is moving with a velocity vA when it is struck by a 2.4-lb ball B which has a velocity vB of magnitude vB = 18 ft/s. Knowing that the velocity of ball B is zero after impact and that...
-
For the beam and loading shown, determine (a) The distance a for which the maximum absolute value of the bending moment in the beam is as small as possible, (b) The corresponding value of M max. 14...
-
The 2015 annual report of the Procter & Gamble Company (P&G) is available at www.pginvestor.com. After reviewing P&Gs annual report, respond to the following questions: a. When does P&G recognize...
-
Consider a publicly held company whose products you are familiar with. Some examples might include: Access the companys public website and search for its most recent annual report. (Some companies...
-
The 2014 financial statements of LVMH Moet Hennessey-Louis Vuitton S.A.LVMH is a Paris-based holding company and one of the worlds largest and best-known luxury goods companies. As a member-nation of...

Study smarter with the SolutionInn App


