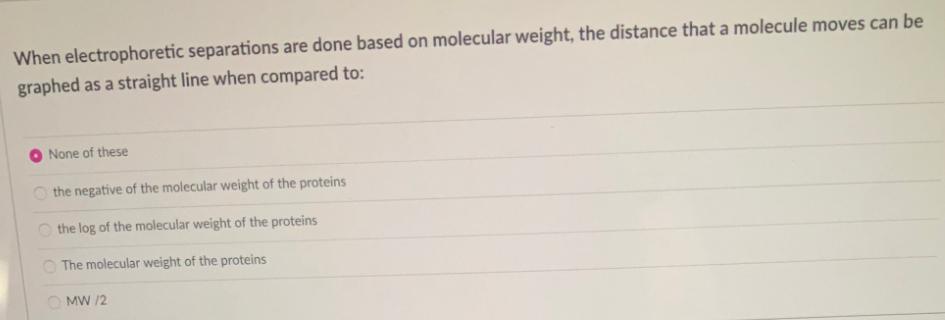
When electrophoretic separations are done based on molecular weight, the distance that a molecule moves can...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
When electrophoretic separations are done based on molecular weight, the distance that a molecule moves can be graphed as a straight line when compared to: None of these O the negative of the molecular weight of the proteins the log of the molecular weight of the proteins O The molecular weight of the proteins O MW /2 In the active site of chymotrypsin Histidine 57 acts as: D in chlorination reactions Da phosphorylation activator in disulfide bond formations O in acid-base reactions Oin zymogen reactions When electrophoretic separations are done based on molecular weight, the distance that a molecule moves can be graphed as a straight line when compared to: None of these O the negative of the molecular weight of the proteins the log of the molecular weight of the proteins O The molecular weight of the proteins O MW /2 In the active site of chymotrypsin Histidine 57 acts as: D in chlorination reactions Da phosphorylation activator in disulfide bond formations O in acid-base reactions Oin zymogen reactions
Expert Answer:
Answer rating: 100% (QA)
Ans1 The molecular weight of the proteins When electrophoretic se... View the full answer

Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Posted Date:
Students also viewed these chemistry questions
-
A 5.0 kg block moves in a straight line on a horizontal frictionless surface under the influence of a force that varies with position as shown in Figure. The scale of the figure's vertical axis is...
-
Chlorination reactions of certain alkanes can be used for laboratory preparations. Examples are the preparation of chlorocyclopropane from cyclopropane and chlorocyclobutane from cyclobutane. What...
-
The imidazole ring of the histidine side chain acts as a proton acceptor in certain enzymecatalyzed reactions. Which is the more stable protonated form of the histidine residue, A or B? Why CH CHC...
-
Generics, Inc. is a U. S. GAAP reporter that manufactures and sells generic drugs and has a December 31 year-end. On March 1, 2014, it began selling a drug, Anocyn, which is a generic of Dicital....
-
Construct a network activity diagram based on the following information: Activity Preceding Activities A .. --- B .. --- C .. A D .. B, C E .. B F .. C, D G .. E H .. F I .. G, H
-
Jayden, a calendar year taxpayer, paid $16,000 in medical expenses and sustained a $20,000 casualty loss in 2021 (the loss occurred in a Federally declared disaster area). He expects $12,000 of the...
-
Consider the heat equation (6.1) with \(a=0.1\) solved in the interval \(0
-
Each of the following internal controls has been taken from a standard internal control questionnaire used by a CPA firm for assessing control risk in the payroll and personnel cycle. 1. Approval of...
-
Replacement Analysis The Everly Equipment Company's flange-lipping machine was purchased 5 years ago for $55,000. It had an expected life of 10 years when it was bought and its remaining depreciation...
-
Complete a failure mode effect analysis for the nitric acid plant reactor section described in Example 9.2. (This is best carried out as a group activity with a group size of three to six). Data from...
-
The following money market account rates were available from a particular bank. Find the compound amount and the interest earned. $6000 at 1.08% compounded daily for one year The compound amount is $...
-
If 1 million people take a standardized test and Sarah scores two standard deviations above the mean, what percentile is she? 50% 68% 0 74% 85% O 97% Save Answer
-
1. Prepare one-page case brief of Marbury v. Madison explaining the power of the Supreme Court's judicial review, including whether the judicial review amounts to law-making. Name of Case Facts of...
-
Problem 5. A certain town has 250,000 families, of which 25,000 do not have a TV at home. As part of an opinion survey, a simple random sample of 900 families is chosen. What is the chance that...
-
The City of Melbourne has created an enterprise fund to provide recreational activities for the community through a city owned health club. Listed below are transactions related to the City of...
-
A highway traffic safety organization collects traffic safety-related data for a certain country. Suppose according to their data, 10,424 fatal collisions in a certain year were the result of...
-
Please show step by step so I can understand. Thanks!Go to the Internet or other sources to gather information for apublicly listed company of yourchoice (i.e., a company listed on a public exchang 2...
-
Horse serum containing specific antibody to snake venom has been a successful approach to treating snakebite in humans. How do you think this anti-venom could be generated? What are some advantages...
-
Oxygen (O2) gas at 273 K and 1.0 atm is confined to a cubical container 10 cm on a side. Calculate Ug/Kavg, where Ug is the change in the gravitational potential energy of an oxygen molecule falling...
-
Three long wires all lie in a xy plane parallel to the x axis. They are spaced equally, 10 cm apart. The two outer wires each carry a current of 5.0 A in the positive x direction. What is the...
-
Figure shows, in cross section, four thin wires that are parallel, straight, and very long. They carry identical currents in the direction indicated. Initially all the four wires are at distance d =...
-
Using the Internet, locate and review these sites: a. National Fraud Center b. The Fraud Detectives Consultant Network c. Milberg Weiss Bershad Hynes & Lerach d. Mark R. Simmons e. Communications...
-
Go to the Internet and peruse the AICPA's "Catch Me If You Can" online fraud game.
-
Search the Internet for the company MAX- IMA Group. List the main consequences of corporate fraud.

Study smarter with the SolutionInn App


