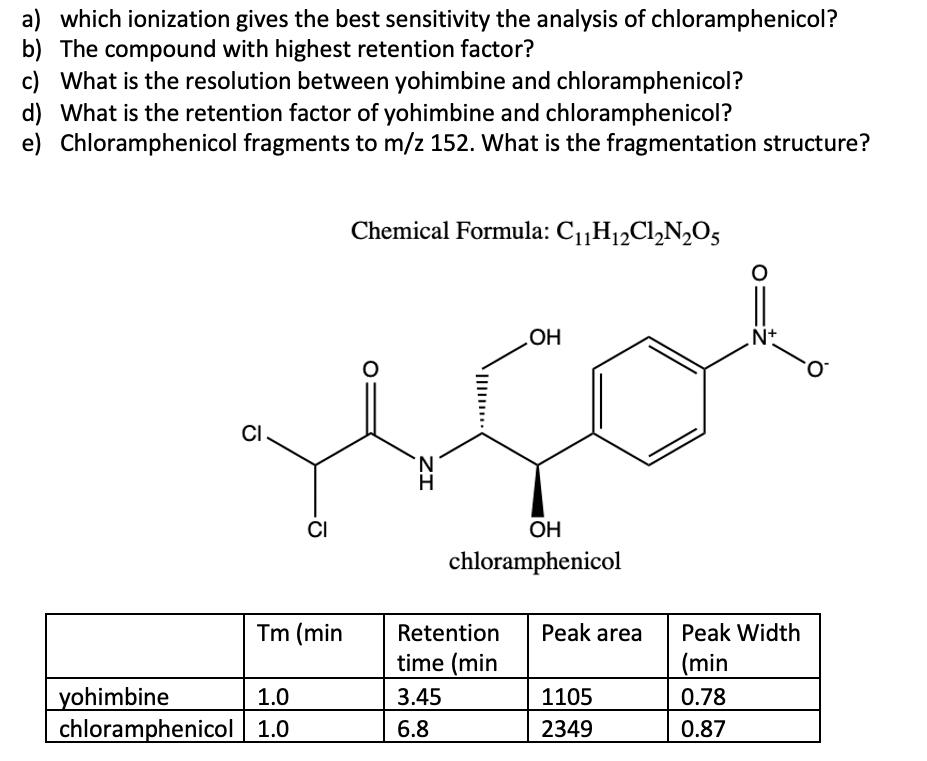
a) which ionization gives the best sensitivity the analysis of chloramphenicol? b) The compound with highest...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
a) which ionization gives the best sensitivity the analysis of chloramphenicol? b) The compound with highest retention factor? c) What is the resolution between yohimbine and chloramphenicol? d) What is the retention factor of yohimbine and chloramphenicol? e) Chloramphenicol fragments to m/z 152. What is the fragmentation structure? Chemical Formula: C1„H12C1,N,O5 HO Nt O. CI. CI OH chloramphenicol Tm (min Retention Peak area Peak Width time (min (min yohimbine chloramphenicol 1.0 1.0 3.45 1105 0.78 6.8 2349 0.87 ZI a) which ionization gives the best sensitivity the analysis of chloramphenicol? b) The compound with highest retention factor? c) What is the resolution between yohimbine and chloramphenicol? d) What is the retention factor of yohimbine and chloramphenicol? e) Chloramphenicol fragments to m/z 152. What is the fragmentation structure? Chemical Formula: C1„H12C1,N,O5 HO Nt O. CI. CI OH chloramphenicol Tm (min Retention Peak area Peak Width time (min (min yohimbine chloramphenicol 1.0 1.0 3.45 1105 0.78 6.8 2349 0.87 ZI
Expert Answer:
Answer rating: 100% (QA)
a The electrospray negative ionization Electrospray Ionization ESI probe in negative ionization mode ... View the full answer

Related Book For 

Applied Statistics in Business and Economics
ISBN: 978-0073521480
4th edition
Authors: David Doane, Lori Seward
Posted Date:
Students also viewed these chemistry questions
-
Compound C (C9H11NO) gives a positive Tollens' test (can be oxidized to a carboxylic acid) and is soluble in dilute HCl. The IR spectrum of C shows a strong band near 1695 cm-1 but shows no bands in...
-
A compound with molecular formula C4H6O gives the infrared spectrum shown in Figure 13.34. Identify the compound. 3.5 4 5 12 13
-
compound with molecular formula C5H10O2 gives the following IR spectrum. When it undergoes acid-catalyzed hydrolysis, the compound with the following 1H NMR spectrum is formed. Identify the...
-
Prove the following statement: If a transformer having a series impedance Ze is connected as an autotransformer, its per-unit series impedance Z as an autotransformer will be SE Zoq NSE + NC Note...
-
What are the principal duties of the Federal Accounting Standards Advisory Board?
-
Dyer and Salinas have decided to form a partnership. They have agreed that Dyer is to invest $120,000 and that Salinas is to invest $40,000. Dyer is to devote one-half time to the business and...
-
The 2002 accounting records of Winona Ryder Co. reveal the following trans- actions and events. Instructions Prepare the cash flows from operating activities section using the direct method. (Not all...
-
Normal Corporation uses standard costing and is in the process of updating its direct materials and direct labor standards for Product 20B. The following data have been accumulated: Direct materials...
-
According to the article "Are Babies Normal?" by Traci Clemons and Marcello Pagano published in The American Statistician, Vol. 53, No. 4, pp. 298-302, the birth weights of babies are normally...
-
In the Bombadier Company, Division A has a product that can be sold either to outside customers or to Division B. Information about these divisions is given below: Case 1 Case 2 Division A: Capacity...
-
Write a function which takes to integer as argument and return there average in float. WAP to test this function. WAP that receives any year from the keyboard and uses a function to determine whether...
-
The goal here is to see if you can develop the intuition for market trends and see how it could impact your business. Please answer each question in paragraph of 6 to 8 lines. Given the disruption to...
-
What is the net payroll tax expenses the company will record in September for the wages of employee Y ? (rounded to nearest dollar) Gross pay through Gross pay for Employee August 31 September X...
-
Discuss the role of effective communication in leadership. How can leaders balance authority with approachability, and what techniques can they use to inspire and motivate their teams while...
-
You need to save a down payment for your house as well as money to cover the property purchase tax and legal fees. You think you will need $50,000 and that you can earn j12=9% on your deposits. How...
-
BBC Company uses the conventional retail inventory method to account for inventory. The following information relates to current - year operations: ( CPA ) Cost . Retail . Beginning inventory &...
-
Degree of Operating Leverage, Percent Change in Profit Ringsmith Company is considering two different processes to make its product-process 1 and process 2. Process 1 requires Ringsmith to...
-
Consider the activities undertaken by a medical clinic in your area. Required 1. Do you consider a job order cost accounting system appropriate for the clinic? 2. Identify as many factors as possible...
-
Engineers are testing company fleet vehicle fuel economy (miles per gallon) performance by using different types of fuel. One vehicle of each size is tested. Does this sample provide sufficient...
-
At a University of Colorado woman's home basketball game, a random sample of 25 con- cession purchases showed a mean of $7.12 with a standard deviation of $2.14. For the next week's home game, the...
-
A manufacturing project has five independent phases whose completion must be sequential. The time to complete each phase is a random variable. The mean and standard deviation of the time for each...
-
The production budget for cars for May (in units) is a. 6,500 b. 11,000 c. 4,500 d. 10,100
-
The total cost of goods sold for June is a. $103,500 b. $128,800 c. 57,500 d. \($232,300\) Munch Enterprises makes a small toy car that is voice activated. Projected sales for the next four months...
-
What is Munchs expected profit for April? a. $192,500 b. $56,000 c. $66,000 d. $59,000

Study smarter with the SolutionInn App


