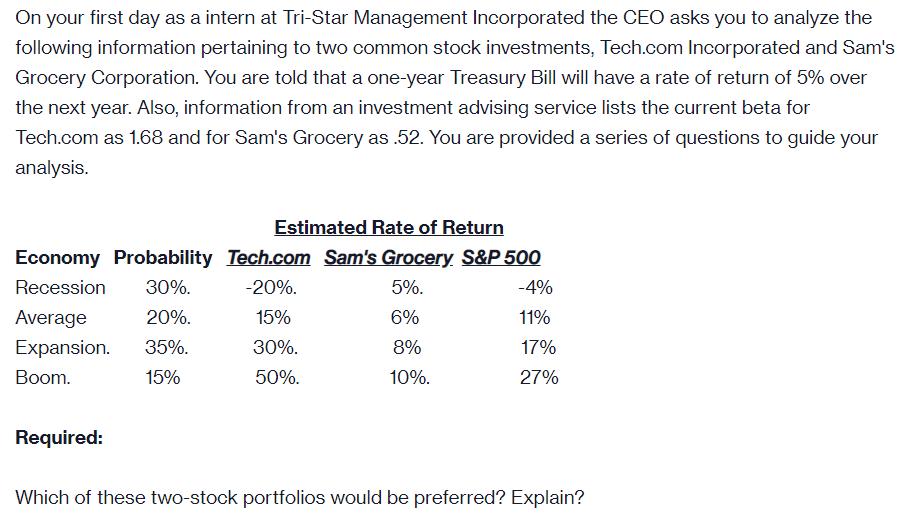
On your first day as a intern at Tri-Star Management Incorporated the CEO asks you to...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
On your first day as a intern at Tri-Star Management Incorporated the CEO asks you to analyze the following information pertaining to two common stock investments, Tech.com Incorporated and Sam's Grocery Corporation. You are told that a one-year Treasury Bill will have a rate of return of 5% over the next year. Also, information from an investment advising service lists the current beta for Tech.com as 1.68 and for Sam's Grocery as .52. You are provided a series of questions to guide your analysis. Estimated Rate of Return Economy Probability Tech.com Sam's Grocery. S&P 500 Recession 30%. -20%. 5%. -4% Average 20%. 15% 6% 11% Expansion. 35%. 30%. 8% 17% Boom. 15% 50%. 10%. 27% Required: Which of these two-stock portfolios would be preferred? Explain? On your first day as a intern at Tri-Star Management Incorporated the CEO asks you to analyze the following information pertaining to two common stock investments, Tech.com Incorporated and Sam's Grocery Corporation. You are told that a one-year Treasury Bill will have a rate of return of 5% over the next year. Also, information from an investment advising service lists the current beta for Tech.com as 1.68 and for Sam's Grocery as .52. You are provided a series of questions to guide your analysis. Estimated Rate of Return Economy Probability Tech.com Sam's Grocery. S&P 500 Recession 30%. -20%. 5%. -4% Average 20%. 15% 6% 11% Expansion. 35%. 30%. 8% 17% Boom. 15% 50%. 10%. 27% Required: Which of these two-stock portfolios would be preferred? Explain?
Expert Answer:
Answer rating: 100% (QA)
To determine which twostock portfolio would be preferred we need to calculate the expected rate of return for each portfolio and then compare them Fir... View the full answer

Related Book For 

Introduction to Corporate Finance What Companies Do
ISBN: 978-1111222284
3rd edition
Authors: John Graham, Scott Smart
Posted Date:
Students also viewed these accounting questions
-
1. Calculate the expected rate of return for Tech.com Incorporated, Sams Grocery Corporation, and the S&P 500 Index. 2. Calculate the standard deviations of the estimated rates of return for Tech.com...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
On your first day as a student marketing intern at the O-Tay Research Company, the supervisor hands you a list of yesterdays telephone interviewer records. She tells you to analyze them and to give...
-
A 35 ft3 rigid tank has air at 225 psia and ambient 600 R connected by a valve to a piston cylinder. The piston of area 1 ft2 requires 40 psia below it to float, Fig. P3.99. The valve is opened and...
-
Distinguish among: cash dividends, property dividends, liquidating dividends, and stock dividends.
-
A random sample of 12-ounce milkshakes from 14 fast-food restaurants had the following number of calories. Assume the population standard deviation is = 90. a. Explain why it is necessary to check...
-
At the beginning of the year, The Claremont Comwae, pany purchased a 100-ton press for \($750,000\). The equipment had an estimated useful life of ten years and a salvage value of \($30,000\). The...
-
Orange Designs provides consulting services related to home decoration. Orange Designs provides customers with recommendations for a full range of home dcor, including window treatments, carpet and...
-
A 4 - year project has an annual operating cash flow of $ 4 8 , 5 0 0 . At the beginning of the project, $ 3 , 9 5 0 in net working capital was required, which will be recovered at the end of the...
-
A village has six residents, each of whom has accumulated savings of $100. Each villager can use this money either to buy a government bond that pays 15 percent interest per year or to buy a year-old...
-
Discuss how the "P" Factors model of assessment can be used to competently and supportively assess the functioning and needs of GSM older adults.?
-
A USDCNH futures contract on HKEX covers USD100,000 with a minimum price change of $0.0001 is sold for CNH 6.6546. If you just place the initial margin of CNH11,305 and the maintenance margin is CNH...
-
Solve the triangle: If B = 100, C = 36.22, b = 5; Find c (round to the nearest whole number) Solve the triangle: If b = 3, a = 4.5, C = 20.74; Find c (round to the nearest whole number) Solve the...
-
When in which condition the promotional goal "to inform" is particularly important? explain.
-
When did Apple first enter the South Korea Market? What was Apple's marketing strategy to penetrate the Korean market? Provide citations.
-
Sarik was surprised by how many conversations it took to make a deal with CorpCo. What pitfall did Sarik experience? Select an answer: using improper anchors burning relationships low-ball...
-
5 4. Find x,y,z,n,m by showing your solution. 2 B D 3 0 1 - S 2 + C E Hops A B C D E F 0 0 8 8 8 8 8 1 0 2 y Z 8 8 2 0 x 1 5 n 10 3 0 1 1 5 6 m 4 0 1 1 5 6 8 F
-
On 1 July 2021, Croydon Ltd leased ten excavators for five years from Machines4U Ltd. The excavators are expected to have an economic life of 6 years, after which time they will have an expected...
-
List and briefly describe the key services investment banks provide to firms before, during, and after a securities offering.
-
Suppose investors face a tax rate of 40 % on interest received from corporate bonds. Suppose AAA-rated corporate bonds currently offer yields of about 7 %. Approximately what yield would AAA-rated...
-
Use the following information to answer the questions that follow. a. Use the DuPont system to compare the two heavy metal companies shown above (HMM and MS) during 2012. Which of the two has a...
-
A packed tower is used to absorb ammonia from air using aqueous sulfuric acid. Gas enters the tower at \(31.0 \mathrm{lbmol} /\left(\mathrm{h}-\mathrm{ft}^{2} ight)\) and is 1.0 \(\mathrm{mol} \%\)...
-
Water originally saturated with carbon tetrachloride \(\left(\mathrm{CCl}_{4} ight)\) at \(25.0^{\circ} \mathrm{C}\) and \(1.0 \mathrm{~atm}\) is stripped with pure air at \(25.0^{\circ} \mathrm{C}\)...
-
We are separating methanol and water in a staged distillation column at total reflux to determine Murphree efficiency. Pressure is \(101.3 \mathrm{kPa}\). The column has a 2.0 -in. head of liquid on...

Study smarter with the SolutionInn App


