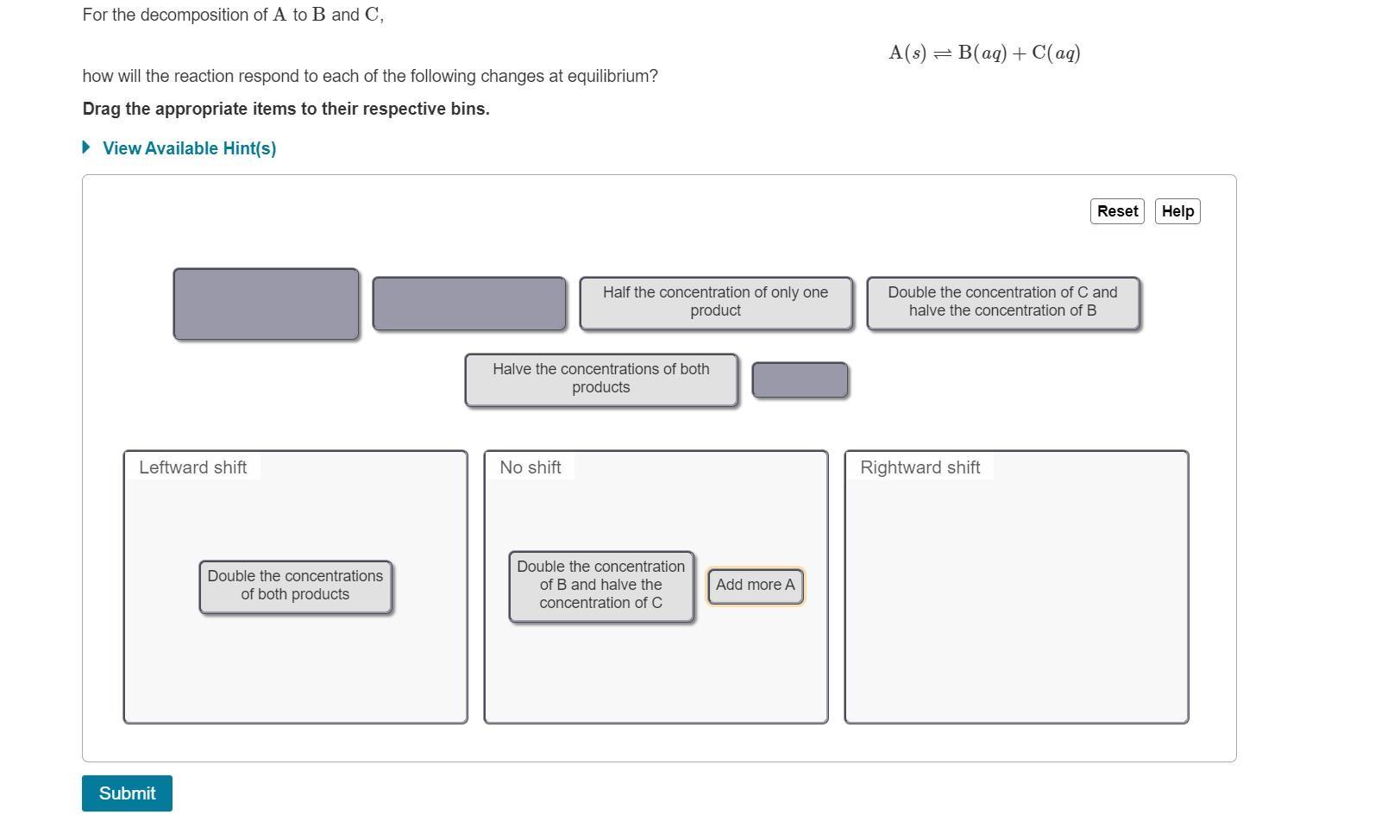
For the decomposition of A to B and C, A(s) = B(aq) + C(aq) how will...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
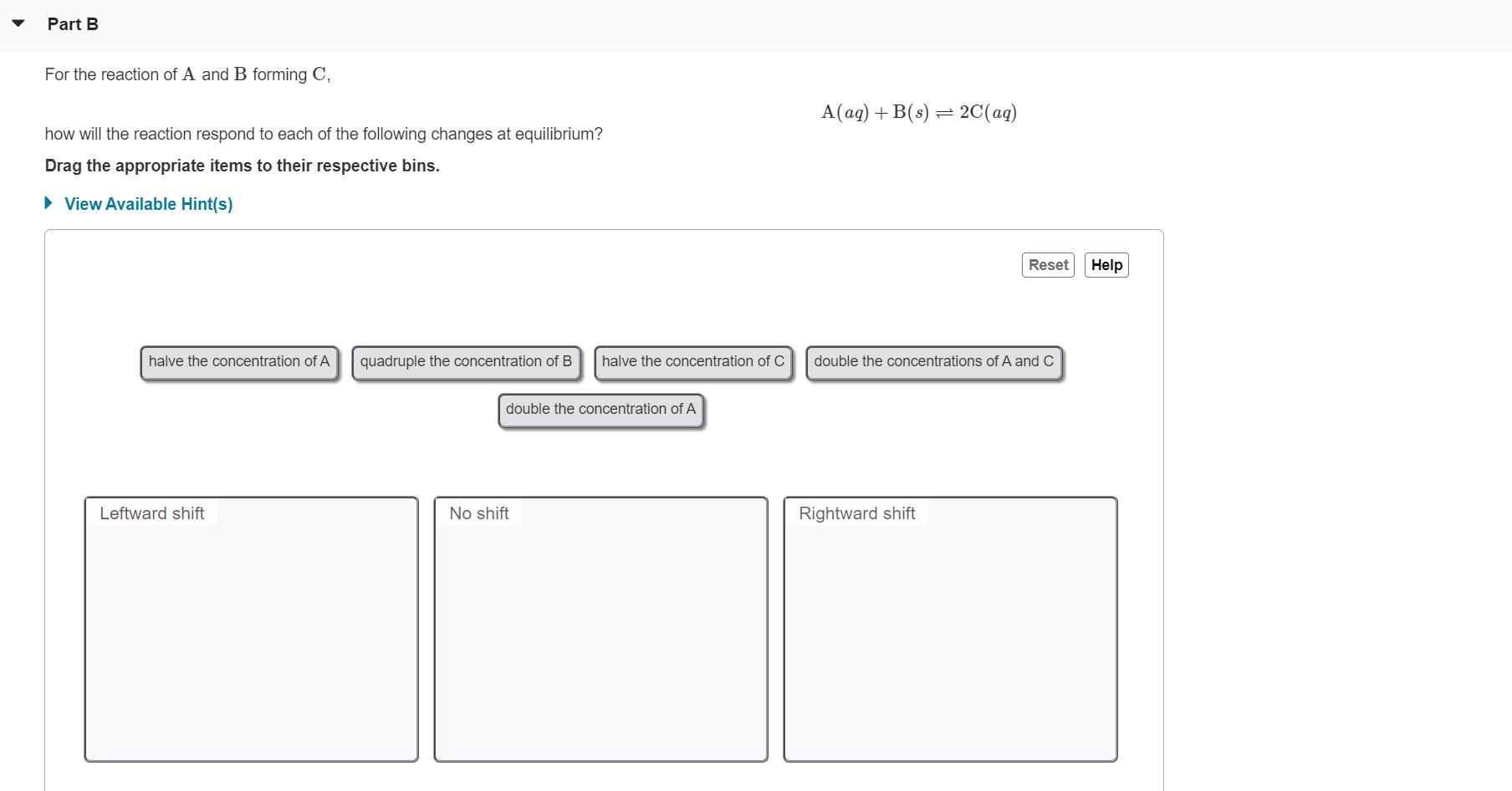
For the decomposition of A to B and C, A(s) = B(aq) + C(aq) how will the reaction respond to each of the following changes at equilibrium? Drag the appropriate items to their respective bins. • View Available Hint(s) Reset Help Half the concentration of only one product Double the concentration of C and halve the concentration of B Halve the concentrations of both products Leftward shift No shift Rightward shift Double the concentration Double the concentrations of B and halve the Add more A of both products concentration of C Submit Part B For the reaction of A and B forming C, A(aq) + B(s) = 2C(ag) how will the reaction respond to each of the following changes at equilibrium? Drag the appropriate items to their respective bins. • View Available Hint(s) Reset Help halve the concentration of A quadruple the concentration of B halve the concentration of C double the concentrations of A and C double the concentration of A Leftward shift No shift Rightward shift For the decomposition of A to B and C, A(s) = B(aq) + C(aq) how will the reaction respond to each of the following changes at equilibrium? Drag the appropriate items to their respective bins. • View Available Hint(s) Reset Help Half the concentration of only one product Double the concentration of C and halve the concentration of B Halve the concentrations of both products Leftward shift No shift Rightward shift Double the concentration Double the concentrations of B and halve the Add more A of both products concentration of C Submit Part B For the reaction of A and B forming C, A(aq) + B(s) = 2C(ag) how will the reaction respond to each of the following changes at equilibrium? Drag the appropriate items to their respective bins. • View Available Hint(s) Reset Help halve the concentration of A quadruple the concentration of B halve the concentration of C double the concentrations of A and C double the concentration of A Leftward shift No shift Rightward shift
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
At 400 K, the half-life for the decomposition of a sample of a gaseous compound initially at 55.5 kPa was 340 s. When the pressure was 28.9 kPa, the half-life was 178 s. Determine the order of the...
-
The rate constant for the decomposition of a certain substance is 1.70 x 10-2 dm3 mol-1 S-1 at 24C and 2.01 X 10-2 dm3 mol-1 S-1 at 37"C. Evaluate the Arrhenius parameters of the reaction.
-
The equilibrium constant Kc for the decomposition of phosgene, COCl2, is 4.63 Ã 10-3 at 527°C: Calculate the equilibrium partial pressure of all the components, starting with pure phosgene...
-
Elastic bands are attached to tuning fork 1 ( which was 2 5 6 Hz ) to reduce its frequency. It is sounded again with tuning fork 2 ( 2 5 5 Hz ) , making 1 2 beats in 6 . 0 s . What is the new...
-
Mattel and Hasbro are two of the largest and most successful toymakers in the world, in terms of the products they sell and their receivables management practices. To evaluate their ability to...
-
Use a calculator to graph all three parabolas on the same coordinate system. Describe (a) the shifts and (b) the stretching and shrinking. (a) y = x 2 (b) y = 3x 2 (c) y = 1/3x 2
-
Suppose you want to organize books onto shelves, and you want the shelves to hold the same number of books. Each shelf will only contain one genre of book. You have 24 sci-fi, 42 fantasy, and 30...
-
The Distance Plus partnership has the following capital balances at the beginning of the current year: Tiger (50% of profits and losses) . . . . . . . . . . . . . . $85,000 Phil (30%) . . . . . . . ....
-
(DuPont analysis) Garwryk, Inc., which is financed with debt and equity, presently has a debt ratio of 84 percent. What is the firm's equity multiplier? How is the equity multiplier related to the...
-
When Pablo Gonzalez died unmarried in 2018, he left an estate valued at $7,850,000. His trust directed distribution as follows: $20,000 to local hospital, $160,000 to his alma mater, and the...
-
Answere the following questions according to the diagram given below Sample Data for Regional Labs ProjectID 100-A 100-A 100-B 200-A 200-B 200-C 200-C 200-D EmployeeName Eric Jones Donna Smith...
-
WHY is Rent Revenue accounted for under Other Income and Expense on the Income Statement?
-
What is the lamina propria and where is it located? Given thathigh number of eosinphils i the lamina propria suggests an allergicreaction, how might that lead to intestinal problems?
-
Great National Limited (GNL), is a multinational company with diverse product ranges and operating in many geographical markets. GNL is finalizing its financial statements for the year ended 30...
-
2. [3 pt] Solve the following recursion exactly by drawing a recursion tree. (You will need to give an exact explicit solution for 7). T(n) = T +T 1 (1/3)+ +n
-
Your client, the Washington Unicorns, is in negotiation with Jordy Michaels for the contract that will cover the final season of his amazing basketball career. Michaels was reasonably healthy last...
-
True or False, The automatic GST exemption allocation for direct skip transfers applies as much of the transferor's remaining GST exemption as is necessary to create a zero inclusion ratio (or as...
-
In Exercises, find the equation of the tangent line at the given point on each curve. 2y 2 - x = 4; (16, 2)
-
Calculate the molar susceptibility of cyclohexane given that its volume susceptibility is -7.9 x 10-7 and its density 811 kg m-3 at 25C.
-
Refer to Fig. 23.3 and determine the pressure range for a branching chain explosion in the hydrogen-oxygen reaction at (a) 700 K, (b) 900 K.
-
50.0 dm3 of dry air was slowly bubbled through a thermally insulated beaker containing 250 g of water initially at 25C. Calculate the final temperature. (The vapour pressure of water is approximately...
-
Decker Corporation experienced a loss in 2020. Additional information for Decker Corporation follows. There were no temporary differences from 2018 to 2020 other than any related to a net operating...
-
Toner Corporation computed the following taxable income and loss: 2020 taxable income, \(\$ 10,000\) and 2021 taxable loss, \(\$ 40,000\). At the end of 2021 , Toner made the following estimates:...
-
DNSE Inc. began operations in 2019. In its first year the company had a net operating loss of \(\$ 10,000\), which was carried forward and used to reduce income tax payable in 2020. In 2020, DNSE had...

Study smarter with the SolutionInn App