Q5: A quantity of 1.4 moles of an ideal gas go through the 3 processes described...
Fantastic news! We've Found the answer you've been seeking!
Question:
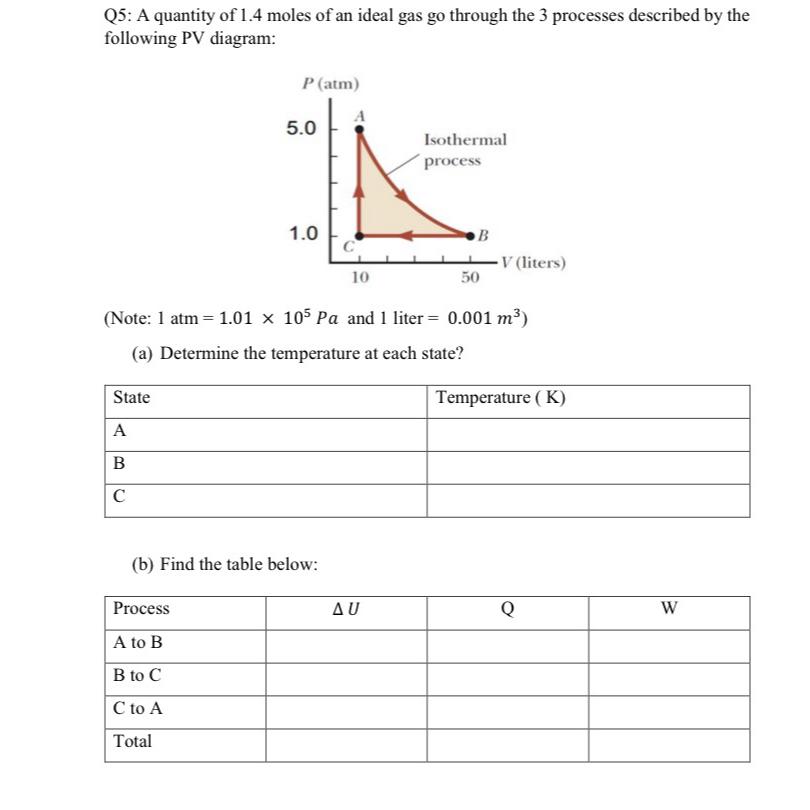
Transcribed Image Text:
Q5: A quantity of 1.4 moles of an ideal gas go through the 3 processes described by the following PV diagram: State A B P (atm) 5.0 Process A to B B to C C to A Total 1.0 (b) Find the table below: C 10 Isothermal process (Note: 1 atm = 1.01 105 Pa and 1 liter = 0.001 m) (a) Determine the temperature at each state? AU B 50 V (liters) Temperature (K) Q W Q5: A quantity of 1.4 moles of an ideal gas go through the 3 processes described by the following PV diagram: State A B P (atm) 5.0 Process A to B B to C C to A Total 1.0 (b) Find the table below: C 10 Isothermal process (Note: 1 atm = 1.01 105 Pa and 1 liter = 0.001 m) (a) Determine the temperature at each state? AU B 50 V (liters) Temperature (K) Q W
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these physics questions
-
Consider now the dynamic power dissipation of different versions of a given processor for three different voltages given in the following table. Determine the static and dynamic power dissipation...
-
What are some examples of an individual's cash flows function as annuities or perpetuities? How do I find the value and future value of each and determine which cash flows give the greatest liquidity...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
You are the president of a new, capital-rich firm and you must decide on your firm's targeting approach. Three large subgroups exist in your market that have never had their unique needs served. If...
-
Write the power series expansions for ei,x. Prove that the real terms give the power series for cosx, while the imaginary terms give that of sin x. Use this identification to justify Euler's formula...
-
Question 5 A leading magazine (like Barron's) reported at one time that the average number of weeks an individual is unemployed is 28 weeks. Assume that the length of unemployment is normally...
-
Total Networks, Inc., reported gross accounts receivable of \($1.2\) billion in 2008 and an allowance for doubtful accounts of \($300\) million. In 2009, the companys gross accounts receivable...
-
Manufacturing cost flow for one-year period Fulton Manufacturing started 2011 with the following account balances. Transactions during 2010 1. Purchased $2,880 of raw materials with cash. 2....
-
G - Why might the risk manager of a corporation interact with the accounting department? H - What type of information would be used in a risk management policy statement?
-
A parent company acquired 70% of the stock of a subsidiary company on January 1, 2017, for $610,000. On this date, the balances of the subsidiarys stockholders equity accounts were Common Stock,...
-
The financial year of Sherlon Ltd ended on 30 June 2015. Your auditors report was signed on 25 August and the financial statements were issued on 10 September. Listed below are events that occurred...
-
Solve the following differential equations. a) dy + 3y = 2xe- ax b) +3x=2t, x(2)=1 dt
-
4. An ideal turbojet engine operates on the Brayton cycle. At the inlet of the compressor (at station 2) the stagnation pressure is 100 kPa and the temperature is 250 K. The compressor pressure ratio...
-
Wilbur and Orville are brothers. They're both serious investors, but they have different approaches to valuing stocks. Wilbur, the older brother, likes to use the dividend valuation model. Orville...
-
Using the STAR framework, give examples of how the below topics contribute to a successful group assignment: 1. Ability to work in a collaborative team environment 2. Demonstrated problem-solving...
-
8. A skydiver is descending with a constant velocity. Consider air resistance. Diagram the forces acting upon the skydiver.
-
Two cars collide at an icy intersection and stick together afterward. The first car has a mass of 1900 kg and was approaching at 13.00 m/s due south. The second car has a mass of 900 kg and was...
-
MgO prevents premature evaporation of Al in a furnace by maintaining the aluminum as Al2O3. Another type of matrix modifier prevents loss of signal from the atom X that readily forms the molecular...
-
The irreversible gas-phase reaction Acat B is carried out adiabatically over a packed bed of solid catalyst particles. The reaction is first order in the concentration of A on the catalyst surface...
-
Read the Web Module Chemical Reaction Engineering of Hippopotamus Stomach on the CRE Web site (http://www.umich.edu/~elements/6e/web_mod/hippo/index.htm). a. Work problems (1) and (2) in the Hippo...
-
Go to the Web site (http://www.umich.edu/~elements/6e/06chap/iclicker_ch6_q1.html) and view at least five i>clicker questions. Choose one that could be used as is, or a variation thereof, to be...
-
A zero coupon bond with a par value of $100 matures in five years. (a) What is the price of the bond if the yield to maturity is 5 per cent? (b) What is the price of the bond if the yield to maturity...
-
Find the present yield to maturity on government securities with maturities of one year, five years and ten years in the Financial Times. How has the yield curve changed since 2010 as shown in the...
-
Bond 1 has an annual coupon rate of 6 per cent and Bond 2 has an annual coupon of 12 percent. Both bonds mature in one year and have a par value of 100. If the yield to maturity on bonds of this risk...

Study smarter with the SolutionInn App


