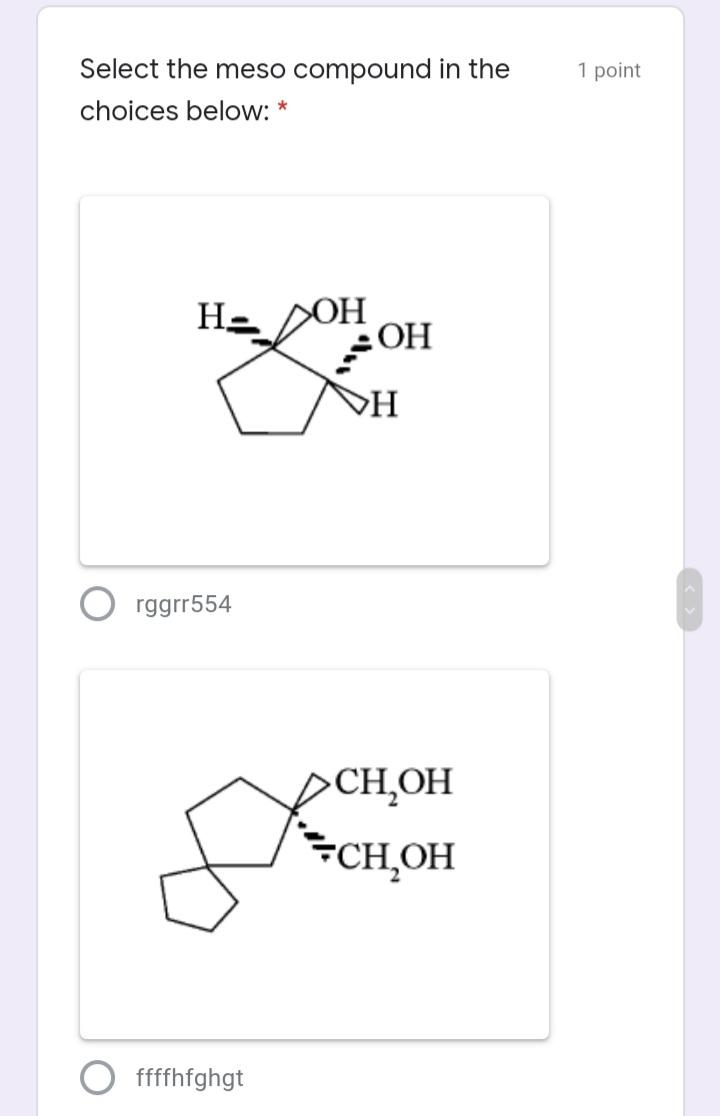
Select the meso compound in the 1 point choices below: * >OH >H rggrr554 CH,OH...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
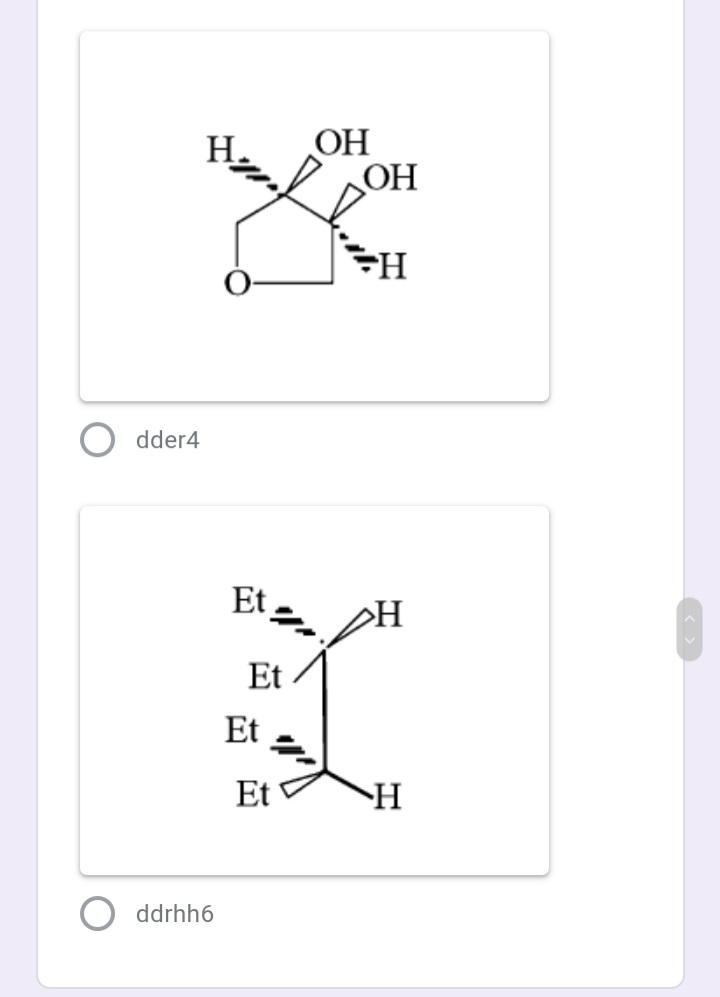
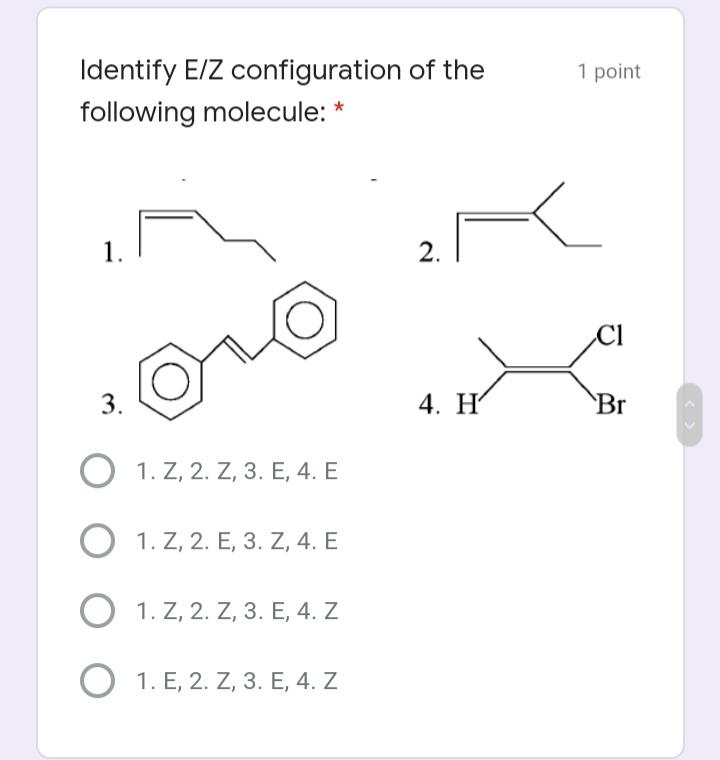
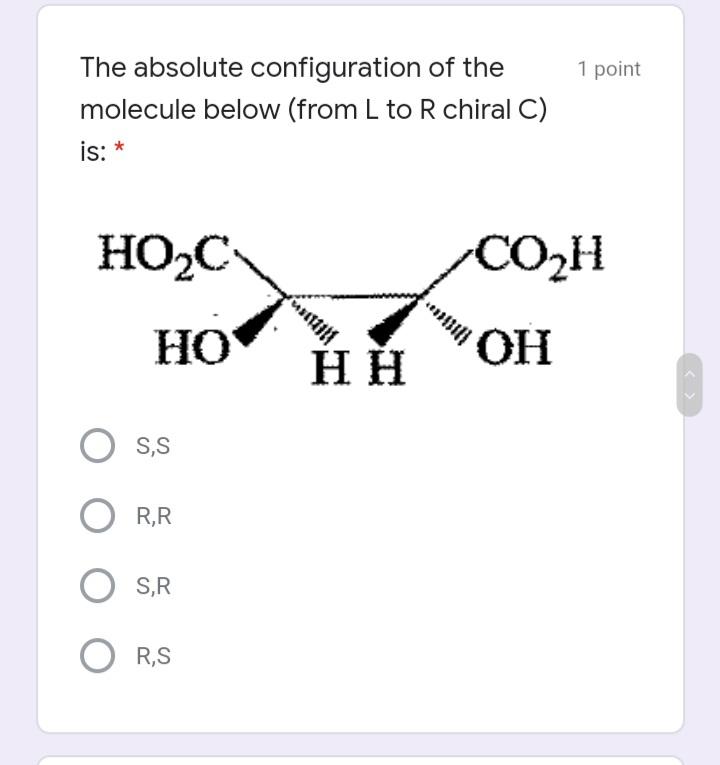
Select the meso compound in the 1 point choices below: * >OH НО >H rggrr554 CH,OH CH,OH ffffhfghgt H. OH OH dder4 Et Et Et Et ddrhh6 Identify E/Z configuration of the 1 point following molecule: 1. 2. 3. 4. Н Br O 1. Z, 2. Z, 3. E, 4. E 1. Z, 2. E, 3. Z, 4. E O 1. Z, 2. Z, 3. E, 4. Z O 1. E, 2. Z, 3. E, 4. Z The absolute configuration of the 1 point molecule below (from L to R chiral C) is: * HO,C CO2H HO н S,S R,R S,R R,S The following compound undergoes 1 point monobromination yielding two products. What is the relationship of the two products? * diastereomers enantiomers meso compounds none of the choices Select the meso compound in the 1 point choices below: * >OH НО >H rggrr554 CH,OH CH,OH ffffhfghgt H. OH OH dder4 Et Et Et Et ddrhh6 Identify E/Z configuration of the 1 point following molecule: 1. 2. 3. 4. Н Br O 1. Z, 2. Z, 3. E, 4. E 1. Z, 2. E, 3. Z, 4. E O 1. Z, 2. Z, 3. E, 4. Z O 1. E, 2. Z, 3. E, 4. Z The absolute configuration of the 1 point molecule below (from L to R chiral C) is: * HO,C CO2H HO н S,S R,R S,R R,S The following compound undergoes 1 point monobromination yielding two products. What is the relationship of the two products? * diastereomers enantiomers meso compounds none of the choices
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What is the shear capacity of the RC beam described below considering the steel reinforcement and using the formula: VRsyAw 2fyd cot 8/s The shear reinforcement in the beam is provided by sets of...
-
Identify whether each of following pairs of compounds are enantiomers or diastereomers: a. b. c. d. e. f. OH OH .CI CI
-
Compound A has two stereoisomers, but compounds B and C exist as single compounds. Explain. CH CH CH CH2 HCH-CH2 CH2CH3 CH3
-
= 0.5. The p.d.f. for the waiting time X until an event occurs often follows the exponential distribution (to be studied in Section 7.6), with the form g(x) = e-x for some positive value of , defined...
-
For the following data: (a) Find the mean, variance, and standard deviation. (b) Construct a frequency table with five classes. (c) Using the grouped data formula, find the mean, variance, and...
-
This assignment is based on the cumulative spreadsheet assignment for Chapter 13. Review that assignment for a summary of the assumptions made in preparing a forecasted balance sheet, income...
-
On January 1, 2000, GAZ Ltd. purchased a producing oil well, with an estimated life of 15 years, and started operating it immediately. The management ofGAZ Ltd. calculated the present value of future...
-
The unadjusted trial balance of Aurora Air Purification System at December 31, 2016, and the data needed for the adjustments follow. Adjustment data at December 31 follow: a. On December 15, Aurora...
-
You invest $1,000 today and expect to sell your investment for $2,000 in 10 years. a. Is this a good deal if the discount rate is 5%? (Do not round intermediate calculations. Round your final an...
-
Select all that apply Actual output is used for variance calculations because Blank______. Multiple select question. budgeted output although useful for planning is outdated for analyzing performance...
-
The ______ is the most common used measure of variability. a. standard deviation b. maturation c. regression d. median
-
Values are standards of conduct. Values are used for judging the goodness or badness of some action. Intrinsic value is something that has value in and of itself (e.g., compassion, honesty,...
-
Successful implementation of the global and the transnational approaches requires high levels of coordination and rapid information flows between corporate headquarters and subsidiaries. Accordingly,...
-
Characterize the current business strategies the company appears to be following with each of its four existing businesses. Form a group with three or four of your classmates. Your group represents...
-
Think of at least two other businesses that are related to the business you will keep and that might be targets for acquisition. Form a group with three or four of your classmates. Your group...
-
Identify existing competitors for the business you chose to keep, including both domestic and international firms. Form a group with three or four of your classmates. Your group represents the...
-
Problem 1 On June 1, 2013, Everly Bottle Company sold $2,000,000 in long-term bonds for $1,754,200. The bonds will mature in 10 years and have a stated interest rate of 8% and a yield rate of 10%....
-
2. In the circuit given in Figure 2, i,(t) = 5.67cos(5t)A and v (t) = 70.71 cos(5t 60) V a) Find the equivalent load impedance. State whether the load is inductive or capacitive. b) Calculate the...
-
(a) Propose a mechanism for the following reaction: (b) Use the bond-dissociation enthalpies given in Table 4-2 (page 143) to calculate the value of (H( for each step shown in your mechanism. (The...
-
There is a small portion of the periodic table that you must know to do organic chemistry. Construct this part from memory, using the following steps. (a) From memory, make a list of the elements in...
-
Calculate the pH of the following solutions. (a) 5.00 g of HBr in 100 mL of aqueous solution (b) 1.50 g of NaOH in 50 mL of aqueous solution
-
From the following selected balances of State plc as at 31 December 2008, prepare (j) a detailed trading and profit and loss account for the year ended 31 December 2008 for internal use and (ii) a...
-
The following information has been extracted from the books of account of Rufford plc for the year to 31 March 2006: Additional information: : 1. Administrative expenses include the following items:...
-
From the following balances in the books of Breaker plc you are to draw up (i) a detailed trading and profit and loss account for the year ending 31 March 2004 for internal use, and (ii) a profit and...

Study smarter with the SolutionInn App


