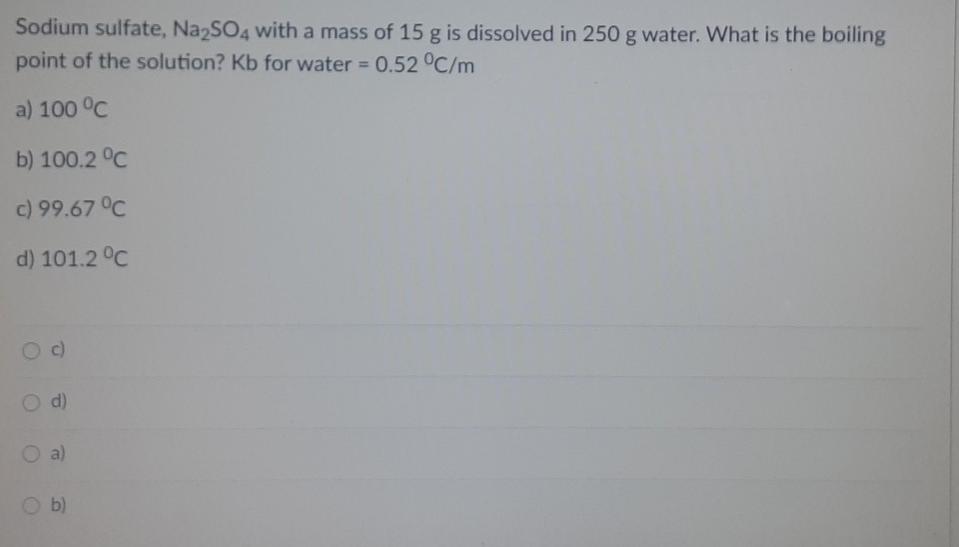
Sodium sulfate, Na2SO4 with a mass of 15 g is dissolved in 250 g water. What...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
Sodium sulfate, Na2SO4 with a mass of 15 g is dissolved in 250 g water. What is the boiling point of the solution? Kb for water = 0.52 °C/m a) 100 °C b) 100.2 °C c) 99.67 °C d) 101.2 °C c) d) O a) b) Fifty grams of an unknown nonelectrolyte solute is mixed with 2000 g of naphthalene (C10H8). The boiling point of the solution was observed to be equal to 80°C. If the boiling point elevation constant of naphthalene is 6.9 °C/m and the boiling point of pure naphthalene is 74.6°C: What is the boiling point elevation of the solution? a) 4 °C b) 5.0 °C c) 5.40 °C d) 5.50 °C a) b) Od) Fifty grams of an unknown nonelectrolyte solute is mixed with 2000 g of naphthalene (C10H8). The boiling point of the solution was observed to be equal to 80°C. If the boiling point elevation constant of naphthalene is 6.9 °C/m and the boiling point of pure naphthalene is 74.6°C: Compute for the molality of the solution. 0.0783 m O 0.783 m 0.0078 m 0.0782 m Sodium sulfate, Na2SO4 with a mass of 15 g is dissolved in 250 g water. What is the boiling point of the solution? Kb for water = 0.52 °C/m a) 100 °C b) 100.2 °C c) 99.67 °C d) 101.2 °C c) d) O a) b) Fifty grams of an unknown nonelectrolyte solute is mixed with 2000 g of naphthalene (C10H8). The boiling point of the solution was observed to be equal to 80°C. If the boiling point elevation constant of naphthalene is 6.9 °C/m and the boiling point of pure naphthalene is 74.6°C: What is the boiling point elevation of the solution? a) 4 °C b) 5.0 °C c) 5.40 °C d) 5.50 °C a) b) Od) Fifty grams of an unknown nonelectrolyte solute is mixed with 2000 g of naphthalene (C10H8). The boiling point of the solution was observed to be equal to 80°C. If the boiling point elevation constant of naphthalene is 6.9 °C/m and the boiling point of pure naphthalene is 74.6°C: Compute for the molality of the solution. 0.0783 m O 0.783 m 0.0078 m 0.0782 m
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What is the boiling point of a solution of 0.150 g of glycerol, C3H8O3, in 20.0 g of water? What is the freezing point?
-
What is the boiling point of a solution made by adding 6.69 g of magnesium chloride to 243.3 g of water? Use the formula of the salt to obtain i.
-
A railroad car having a mass of 15 Mg is coasting at 1.5 m/s on a horizontal track. At the same time another car having a mass of 12 Mg is coasting at 0.75 m/s in the opposite direction. It the cars...
-
Consider the following graph. 2 7 6 C 10 10 H 11 E 12 15 14 (a). Find a minimum spanning tree of the graph using Kruskal's algorithm. List the edges in the order they are put into the tree. (b)....
-
Using the present value tables in Appendix A, calculate the present value of the following: 1. $250,000 to be received three years from today, assuming an annual interest rate of 6%. 2. $2,500 to be...
-
I have a theory that if you ask participants to sort one-sentence characteristics of people (e.g., I eat too fast) into five piles ranging from not at all like me to very much like me, the percentage...
-
Change \(149.11 \times 10^{-4}\) by moving the decimal two places to the right.
-
For each of the following transactions, identify the net asset classification ( unrestricted, temporarily restricted, permanently restricted) that is affected in the NFPOs financial statements for...
-
please do not try to cheat the answer from any other platform , I need your answer. Music Teachers, Inc., is an educational association for music teachers that has 19,800 members. The association...
-
1. Estimate the demand for soft drinks using a multiple regression program available on your computer. 2. Interpret the coefficients and calculate the price elasticity of soft drink demand. 3. Omit...
-
What will be printed after running the following program: module Question9; int a = 3, b = 5, c = 1; initial begin b
-
Harris Fabrics computes its plantwide predetermined overhead rate annually on the basis of direct labor - hours. At the beginning of the year, it estimated that 70 , 0 0 0 direct labor - hours would...
-
Pop Corporation acquired 70 percent of Soda Company's voting common shares on January 1, 202, for $118,300. At that date, the honcontrolling interest had a fair value of $50,700 and Soda reported...
-
Suppose the mass of ice, 1 3 g , is measured with a scale that measures to 0 . 1 g . Calculate the percent precision ( % ) of the measured mass of the ice.
-
Discuss and describe transactional leadership in term of management. ?
-
You bought a share of 4 percent preferred stock for $95.90 last year. The market price for your stock is now $97.38. The par value is $100.What is your total return for last year?
-
Review the "OWLC Plagiarismand Writing Integrity" and other resources in this unit's Overview, Readings & Resources. They include topics on "typical"plagiarism violations as well as self-plagiarism....
-
When an electric field is applied to a shallow bath of vegetable oil, why do tiny bits of thread floating in the oil align with the field like compasses in a magnetic field?
-
What is wrong with the following "proof" that every matrix with at least two rows is row equivalent to a matrix with a zero row? Perform R2 + R1 and R1 + R2. Now rows 1 and 2 are identical. Now...
-
Suppose that A is a 6 6 matrix with characteristic polynomial cA() = (1 + ) (1 - )2 (2 - )3. a. Prove that it is not possible to find three linearly independent vectors v1, v2, v3 in R6 such that...
-
In Exercises 1-3, a matrix A is given along with an iterate x5, produced as in Example 4.30. (a) Use these data to approximate a dominant eigenvector whose first component is 1 and a corresponding...
-
Find the Panel on Audit Effectiveness report by the Public Oversight Board (August 31, 2000) and outline the panel's major recommendations concerning forensic techniques (pp. 1 and 2).
-
Suppose you are fired because you would not "cook the books." Do you have any regress? Check out some whistle-blowers websites, especially the Office of Special Counsel.
-
Find websites describing "big bath" situations.

Study smarter with the SolutionInn App



