Lithium hydride, LiH, has been proposed as a possible nuclear fuel. The nuclei to be used and
Question:
Lithium hydride, LiH, has been proposed as a possible nuclear fuel. The nuclei to be used and the reaction involved are as follows: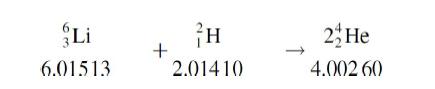
the listed masses being those of the neutral atoms. Calculate the expected power production, in megawatts, associated with the consumption of 1.00 g of LiH per day. Assume 100 percent efficiency.
Transcribed Image Text:
Li 6.01513 + H 2.01410 2 He 4.002 60
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
Ignoring the electron binding energies the change in mass for ...View the full answer

Answered By

Cristine kanyaa
I possess exceptional research and essay writing skills. I have successfully completed over 5000 projects and the responses are positively overwhelming . I have experience in handling Coursework, Session Long Papers, Manuscripts, Term papers, & Presentations among others. I have access to both physical and online library. this makes me a suitable candidate to tutor clients as I have adequate materials to carry out intensive research.
4.90+
1538+ Reviews
3254+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
Read the case study and answer the question below with a one page response. What does a SWOT analysis reveal about the overall attractiveness of Under Armours situation? Founded in 1996 by former...
-
The large magnetic fields used in MRI can produce forces on electric currents within the human body. This effect has been proposed as a possible method for imaging biocurrents flowing in the body,...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
State the issues presented to the Court in McCord v. McCord .
-
The Cooper Electronics Company has developed the following schedule of potential investment projects that may be undertaken during the next six months: a. If Cooper requires a minimum rate of return...
-
The file P12_24.xlsx contains the daily closing prices of Procter & Gamble stock for a one-year period. a. Use only the 2003 data to estimate an appropriate auto regression model. b. Next, use the...
-
The notes to financial statements present information on significant accounting policies, complex or special transactions, details of reported amounts, commitments, contingencies, segments, quarterly...
-
Espinosa Industries is considering an investment in equipment that will replace direct labor. The equipment has a cost of $615,000 with a $75,000 residual value and a 10-year life. The equipment will...
-
You own 100 shares of stock of unlevered Shaan Company which has 1,000 shares outstanding. Shaan plans to pay $2,200 dividend at the end of the current year (i.e., one year from today) and a...
-
A locomotive moving at 30.0 m/s approaches and passes a person standing beside the track. Its whistle is emitting a note of frequency 2.00 kHz. What frequency will the person hear (a) As the train...
-
Neutrons produced by fission reactions must be slowed by collisions with moderator nuclei before they are effective in causing further fissions. Suppose an 800-keV neutron loses 40 percent of its...
-
Assume that the stock of Warner-Lambert Company, a pharmaceutical manufacturer, is currently selling for $75 per share. You have $7,500 of your own funds to invest. The brokerage firm that you use...
-
If \(A x^{2}+C y^{2}+D x+E y+F=0\), for Problems 9-13, then list conditions on the constants to assure that the indicated graph results. an ellipse \(A\) and Chave the same signs
-
In Griffin v. California (1965), a case involving the rape and murder of a woman in a dark alley, the Supreme Court considered whether the following arguments by the prosecutor constituted...
-
Consider the model \[ y=\theta_{1}-\theta_{2} e^{-\theta_{3} x}+\varepsilon \] This is called the Mitcherlich equation, and it is often used in chemical engineering. For example, \(y\) may be yield...
-
Suppose the probabilities are 0.89,0.09, and 0.02 that the finish on a new car will be rated acceptable, easily repairable, or unacceptable. Find the probability that, among 20 cars painted one...
-
The probabilities that a quality control team will visit \(0,1,2,3\), or 4 production sites on a single day are 0.15,0.22,0.35,0.21, and 0.07. (a) Simulate the inspection team's visits on 30 days....
-
1. What specific conduct of Robinson was the state trying to stop? 2. Why did the state supreme court agree that Robinson was not engaging in the unauthorized practice of law by representing others...
-
Cassandra Casey operates the Futuristic Antique Store. She maintains subsidiary ledgers for accounts payable and accounts receivable. She presents you with the following information for October 2019:...
-
The entropy of the working fluid of the ideal Carnot cycle (increases, decreases, remains the same) during the isothermal heat rejection process.
-
Steam is accelerated as it flows through an actual adiabatic nozzle. The entropy of the steam at the nozzle exit will be (greater than, equal to, less than) the entropy at the nozzle inlet.
-
Steam enters a steady-flow adiabatic nozzle with a low inlet velocity as a saturated vapor at 6 MPa and expands to 1.2 MPa. (a) Under the conditions that the exit velocity is to be the maximum...
-
Hi Bo, I'm just trying to budget for our sales team bonus, but I want it to be based on when sales are recognised in the financial statements so that I have a sound basis for the calculation. If we...
-
The following factor(s) affect the length of the operating cycle: Question 9Answer a. Speed of the production process b. Efficiency of ordering and purchasing procedures c. All of them d. Whether...
-
Annie's Homemade Ice Cream is preparing a planning budget for August. Its cost formula for salaries and wages includes managers* salaries of $7,000 per month plus employee wages of $8.00 per...

Study smarter with the SolutionInn App


