A vapor mixture of equal volumes NH 3 and N 2 is contacted at 20 C
Question:
A vapor mixture of equal volumes NH3 and N2 is contacted at 20οC and 1 atm (760 torr) with water to absorb some of the NH3. If 14 m3 of this mixture is contacted with 10 m3 of water, calculate the % of ammonia in the gas that is absorbed. Both T and P are maintained constant. The partial pressure of NH3 over water at 20οC is:
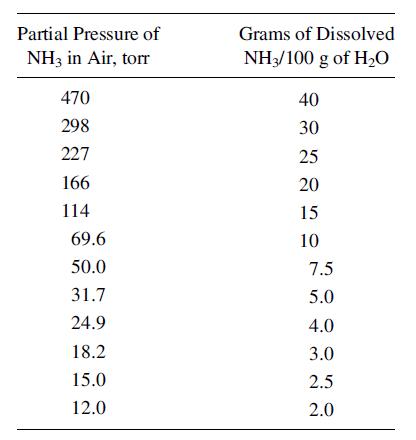
Transcribed Image Text:
Partial Pressure of NH3 in Air, torr 470 298 227 166 114 69.6 50.0 31.7 24.9 18.2 15.0 12.0 Grams of Dissolved NH3/100 g of H₂O 40 30 25 20 15 10 7.5 5.0 4.0 3.0 2.5 2.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
The of ammonia in the gas that is absorbed 14 m3 of gas 1410 m3 of wat...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Separation Process Principles Chemical And Biochemical Principles
ISBN: 9780470481837
3rd Edition
Authors: By J. D. Seader, Ernest J. Henley, D. Keith Roper
Question Posted:
Students also viewed these Life Sciences questions
-
The volume of chlorine at 20 C and 1 atm of pressure is 150 L. What volume will it occupy at 60 C and 2770 mmHg of pressure?
-
What volume of O2 at 20 C and 1 atm of pressure can be obtained from 100 g of KClO3?
-
A vapor mixture of n-butane (B) and n-hexane (H) contains 50.0 mole% butane at 120C and 1.0 atm, A stream of this mixture flowing at a rate of 150.0 L/s is cooled and compressed, causing some but not...
-
Data-2-Go manufactures and sells flash drives. The company produces only when it receives orders and, therefore, has no inventories. The following information is available for the current month:...
-
An investigator wants to take an unequal-probability sample of 10 of the 25 psus in the population listed below and in file exercise0602.dat, and wishes to sample units with replacement. a. Adapt the...
-
Home heating oil is sold by the gallon. Last winter, the Romano family used 370 gallons of oil at a price of $3.91 per gallon. If the price increases 9% next year, what will their approximate heating...
-
Coca-Cola is considered to have one of the most valuable brand names in the world. The firm has an after-tax operating margin of 20% on revenues of \($25\) billion. The capital invested in the firm...
-
The beginning inventory at Keats Office Supplies and data on purchases and sales for a three-month period are as follows: Instructions 1. Based the inventory, purchases, and cost of merchandise sold...
-
On a typical day, a company writes 88 checks worth a total of $10,860 that clear in 2.75 days. The company also collects 101 checks worth a total of $14,875 that clear in 2.25 days. Is this a...
-
Write stubs for all the constructors and methods in the Student class of Figures 8.2a and 8.2b. Each stub should print the constructor or method name followed by the passed-in values of all...
-
Fifty mol% propylene in propane is separated with silica gel. The products are to be 90 mol% propylene and 75 mol% propane. If 1,000 lb of silica gel/lbmol of feed gas is used, can the desired...
-
A colored substance (B) is removed from a mineral oil by adsorption with clay particles at 25C. The original oil has a color index of 200 units/100 kg oil, while the decolorized oil must have an...
-
What is an Actor and what is its relationship to a UC?
-
Salaries represent the costs of employing two new machine operators at a salary of $40,000 per annum each. For the HCC machine, the company will only need to employ a new machine operator and the...
-
Compare and contrast the use of job evaluation methodologies and market pricing to determine salary levels. What are the advantages and disadvantages of each, and when would each be appropriate?...
-
Grant & Krewstown Inc. also records $420,000 in Net Income and pays dividends of $14,000. Assuming this is the company's first year of operations, what is Total Stockholders' Equity? 1000
-
Year 0 1 2 3 4 5 6 Cash Flow -$9,000 $2,000 $3,600 $2,700 $2,100 $2,100 $1,600 A. Payback. The company requires all projects to payback within 3 years. Calculate the payback period. Should it be...
-
What are social responsibility and ethics as they relate to business-oriented organizations
-
Income statement data for Yellowstone Images Inc. for the years ended December 31, 20Y5 and 20Y4, are as follows: a. Prepare a comparative income statement with horizontal analysis, indicating the...
-
Using a graphing utility, graph y = cot -1 x.
-
Many viruses, including WNV, cold viruses, and flu viruses, reproduce in the host for a short period of time before being destroyed by the hosts immune system. This production of new virus particles...
-
In the acquired immune response, two types of lymphocytes, B cells and T cells, are the key players. a. Compare and contrast the antigen receptors of B and T cells. b. B and T cells can only...
-
Each tassel produces 25 million pollen grains. One acre of a cornfield may contain 20,000 to 30,000 corn plants, producing up to 68 kg (approximately 150 pounds) of pollen in a single growing season....
-
You are given an array arr of size N and you are provided with a number K. You have to append K elements from the end of the array to the beginning of the array and print the modified array. Input...
-
The plan assets set aside by the employer are a key element in Multiple choice question. a defined benefit pension plan. a defined contribution pension plan
-
P&P Skateboards had net sales of $2,600,000, its cost of goods sold was $1,700,000, and its net income was $800,000. Its gross margin ratio equals: OOOOO 20 47%. 35%. 31%. 65%. 153%.

Study smarter with the SolutionInn App


