Prove that Equation (15.6) reduces to the LorenzLorentz equation (15.4) in the case of purely ionic bonding.
Question:
Prove that Equation (15.6) reduces to the Lorenz–Lorentz equation (15.4) in the case of purely ionic bonding.
Equation (15.6)
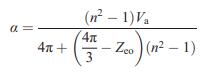
Equation (15.4)
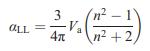
Transcribed Image Text:
a = 4x + (n - 1) Va 4 3 -Zeo (1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Purely ionic bonding implie...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
To calculate variance and standard deviation, we take the deviations from the mean. At times, we need to consider the deviations from a target value rather than the mean. Consider the case of a...
-
A registered dealer, based in Chandigarh, makes a supply to another registered dealer located in Chandigarh, valuing rupees 1,20,000. The applicable rate of GST is 12%. Calculate the amount of tax...
-
In aqueous solution, hydrogen sulfide reduces (a) Fe3+ to Fe2+ (b) Br2 to Br- (c) MNO4- to Mn2+ (d) HNO3 to NO2. In all cases, under appropriate conditions, the product is elemental sulfur. Write a...
-
Suppose that the probability that a mechanic fixes a car correctly is 0.9. Determine the odds against the mechanic fixing a car correctly.
-
Calculate the Lande g factor for atoms (a) In S states; (b) In singlet states.
-
Discuss the advantages and disadvantages of Web-based questionnaires.
-
Go to the Ernst \& Young Web site (https://www. ey.com/Publication/vwLUAssets/ey-worldwide-transfer-pricing-reference-guide-2018-19/\$FILE ley-worldwide-transfer-pricing-reference-guide-2018-19.pdf)...
-
The profitable Palmer Golf Cart Corp. is considering investing $300,000 in special tools for some of the plastic golf cart components. Executives of the company believe the present golf cart model...
-
No, material requisition does not serve the same purpose as a PO. While a PO consist of purchasing from a vendor/supplier detailing the items (materials/service) purchased with detailes such as...
-
Use the AndersonEggleton relationship to calculate the refractive index of the mineral orthoclase (KAlSi 3 O 8 ) with a unit-cell volume 720.4 3 containing four formula units. The cation...
-
Yellow light of the Na doublet with a wavelength of 589.30.3 nm and frequency of 5.0910 14 Hz in vacuum enters Fe 2 O 3 that has a refractive index of 3.00. Calculate the speed, wavelength, and...
-
On April 29, 2015, Auk Corporation acquires 100% of the outstanding stock of Amazon Corporation (E & P of $750,000) for $1.2 million. Amazon has assets with a fair market value of $1.4 million (basis...
-
The space age bicycle rim in Figure 4.88 can be modeled as a thin disk with four circular cutouts, which are of identi- cal size. The density of the disk material is kg/m2 . In Figure 4.88, the rim...
-
The Company produces organic butter for sale. No direct materials other than milk are used. Production data for April 2 0 1 5 are shown below: April Beginning Work in Process Inventory: Units (...
-
Determine the size of the T beam with the specifications shown below with the smallest possible section that can carry the bending moment in the simply supported beam. The maximum allowable stress is...
-
Critically assess how Toyota embeds approaches to quality management and organizational sustainability to create business excellence in the industry. Use the European Foundation for Quality...
-
Flint Company leased equipment to Land Company for a ve-year period. Flint paid $9,393 for the equipment, which equals its current carrying value (with estimated useful life of ve years). The lease...
-
You have been asked by your accounting professor to prepare a paper outlining the importance of the statement of cash flows, the details of what is included in each of the three sections of the...
-
During the month, services performed for customers on account amounted to $7,500 and collections from customers in payment of their accounts totaled $6,000. At the end of the month, the Accounts...
-
An alkyne with molecular formula C 5 H 8 was treated with sodium in liquid ammonia to give a disubstituted alkene with molecular formula C 5 H 10 . Draw the structure of the alkene.
-
Predict the major product expected for each of the following reactions: (a) (b) (c) (d) (e) (f) ? XS HCI CI 1) xs NANH/NH3 2) H,0 CI
-
Suggest reagents that would achieve the following transformation: CI CI CI
-
Following information about an existing bond is available: Principal Time to maturity ( in years ) Annual coupon ( $ ) Price ( $ ) 1 0 0 1 0 9 6 Additionally, assume that the zero rate for two years...
-
Michael received a Schedule K-1 from Omega Partnership reflecting a $25,000 share of ordinary income and an $18,500 cash distribution. As a result of his ownership of Omega, Michael's taxable income...
-
FINANCE QUESTION : Small town Diners has a policy of treating dividends as a passive residual. It forecasts that net earnings after taxes in the coming year will be $500,000. The firm has earned the...

Study smarter with the SolutionInn App


