Predict the spin spin splitting that you would expect to observe in the NMR spectra of
Question:
Predict the spin – spin splitting that you would expect to observe in the NMR spectra of each compound in Problem 37.
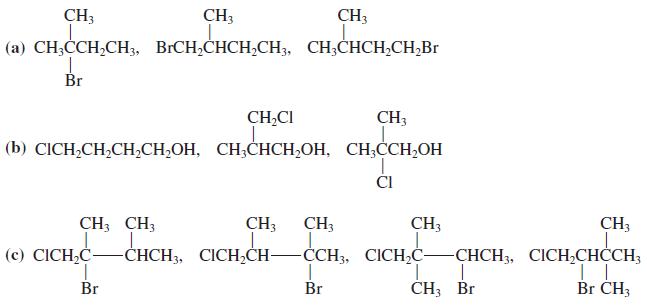
Transcribed Image Text:
CH3 CH3 CH3 (a) CH;CCH,CH3, BICH,CHCH,CH3, CH;CHCH,CH;Br Br CH,CI CH; (b) CICH,CH,CH,СH,ОН, СН,СНСH,ОH, СH,CCH,ОН 1. ČI CH; CH; CH3 CH; CH3 CH3 CH. (с) CICH-C- -СНCH, CICH,CH- -ČCH3, CICHC -СНCH, CICH,СНСCH, Br Br CH3 Br Br CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
SNo spinspin splitting1HNMR 13CNMR a C1s6H C3q2H C4t3H C1q C2s C3t C4q C1d2H C2m1H C2d3H C...View the full answer

Answered By

Bharat Singh Patel
I like teaching since my matriculation. That time i was teaching 6-8 class students with all subjects. I was very interested in teaching. Then after i have done my 10+2. I joined a coaching class. Where i was teaching mathematics and chemistry of 10th class student. After that i moved to Ewing Christian College, Allahabad for Graduation. then i taught chemistry only. then I clear IIT JAM to got admission in IIT Jodhpur for M.Sc. During M.Sc I have cleared CSIR-JRF With AIR 54 and GATE with AIR 114. In college time I always cleared doubts of classmates and juniors.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the products that you would expect to obtain when the following compounds are treated with ozone, followed by work-up with a. Zn, H2O: b. H2O2 1. 2. CH2¡CHCH2CH2CH2CH3 3. 4. 5....
-
Identify two types of companies that you would expect to have a high gross profit margin and two types of companies that you would expect to have a low gross profit margin.
-
Name one element that you would expect to exhibit bonding properties similar to boron. Explain?
-
Write C++ statements to do the following. a. Declare int variables num1 and num2. b. Prompt the user to input two integers. c. Input the first number in num1 and the second number in num2. d. Output...
-
Explain the contents of the procurement management plan.
-
Given a normal distribution centered at 0 with a standard deviation of 1. List the following probabilities in order from smallest to greatest: a.) P(z > 0.5) b.) P(-0.3
-
Construct your key relations chart for this chapter.
-
Bennett Construction Company is a building contractor specializing in small commercial buildings. The company has the opportunity to accept one of two jobs; it cannot accept both because they must be...
-
elaborate!!! *What security policies and physical security measures has an organization implemented? Are they cumbersome to use? Are they effective in protecting the network and its data? *What types...
-
Blanche Devereaux is a 78-year-old widow who was brought to the emergency room one evening by her sister. Earlier in the day, she had seen bright red blood in her stool. She continued with her daily...
-
Predict the spin spin splitting that you would expect to observe in the NMR spectra of each compound in Problem 36. (Reminder: Hydrogens attached to oxygen and nitrogen do not normally exhibit spin ...
-
1 H NMR spectra C through F (see below) correspond to four isomeric alcohols with the mole cular formula C 5 H 12 O. Try to assign their structures. 1 MR 6 H 3 H 'H NMR 6 H 1 H 2 H 1.5 1.4 1.3 0.9...
-
What are four types of economic systems? Can you provide an example of a country using each type?
-
What is Configuration Status Accounting (CSA)? Give examples.
-
What is the difference between a test of independent means and a test of dependent means, and when is each appropriate?
-
When is verification started and when does it end? At what levels of abstraction are V&V performed?
-
What questions should you ask vendors that may have products or services you are considering selecting?
-
Find a book or web site that defines a set of rules for English grammar or the grammar of another human language. Express all of the rules in BNF and as transition networks. What problems do you...
-
Describe in your own words the three strengthening mechanisms discussed in this chapter (i.e., grain size reduction, solid-solution strengthening, and strain hardening). Explain how dislocations are...
-
Aztec Furnishings makes hand-crafted furniture for sale in its retail stores. The furniture maker has recently installed a new assembly process, including a new sander and polisher. With this new...
-
Calculate the pressure exerted by 1.0 mol H2S behaving as (a) A perfect gas, (b) A van der Waals gas when it is confined under the following conditions: (i) At 273.15 K in 22.414 dm3, (ii) At 500 Kin...
-
Express the van der Waals parameters a = 1.32 atm dm6 mol? And b = 0.0436 d3 mol-1 in SI base units.
-
A gas at 350 K and 12 atm has a molar volume 12 per cent larger than that calculated from the perfect gas law. Calculate (a) The compression factor under these conditions and (b) The molar volume of...
-
Suppose the CAPM holds. The risk-free rate is 4% and the expected return of the market portfolio is 10%. You form a portfolio, and the expected return of the portfolio is 16%. Find out the beta of...
-
I need help with some more information on this topic. I am unsure how to access the library articles, and, to be honest, I am overwhelmed. I also need help with organizing. It needs to be about ten...
-
Gross Margin Income Statement Rebound Therapy Inc ("RTI") is a company that provides post-injury, registered massage therapy services for amateur and professional athletes. They also sell a line of...

Study smarter with the SolutionInn App


