Which of the following structures qualify as being aromatic, according to Hckels rule? CH=CH2 (a) A (b)
Question:
Which of the following structures qualify as being aromatic, according to Hückel’s rule?
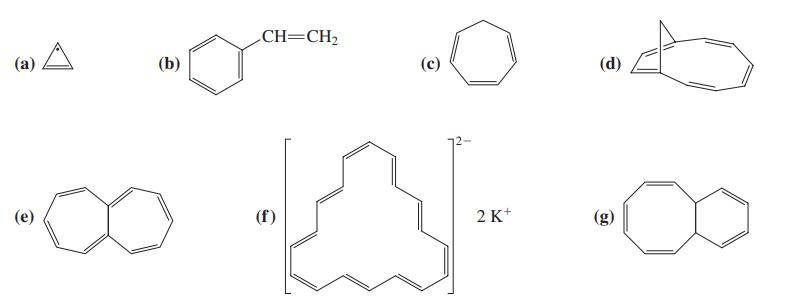
Transcribed Image Text:
CH=CH2 (a) A (b) (с) (d) "00 (е) (f) 2 K*
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (6 reviews)
For a compound to be aromatic according to Huckels rule it shoul...View the full answer

Answered By

Aditi Deswal
Currently , I am doing post graduation ( MSc.) in Chemistry . I want to bacome a lecturer . I feel happy when I teach student and solve their problem . It is my passion as well as hobby . I feel blessed If I got a chance to share my knowledge as much as I have . I teach student at my home also. I love teching and want to spend my whole life to teach the students and explores new things.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Which of the following structures has the smallest heat of combustion? (a) (b) (c) (d) CH3 CH3 CH3 CH3 CH3
-
Which of the following structures represent the same compound? Which ones represent different compounds? (a) (b) (c) (d) (e) (f) (g) Name the structures given in Problem 3-33, parts (a), (c), (e),...
-
Which of the following compounds are aromatic? Are any antiaromatic? NH HN: :NH Nl 1
-
Lamonda Corp. uses a job order cost system. On April 1, the accounts had the following balances: The following transactions occurred during April: (a) Purchased materials on account at a cost of...
-
Differentiate between the systems approach and the systems analysis approach taught in introductory systems analysis and design courses.
-
Bobby Brown has just turned 48 years of age and has come to you seeking retirement advice.During your discussion the following information was communicated: Bobby wants to retire on his 65 th...
-
We are separating methanol and water in a staged distillation column at total reflux to determine Murphree efficiency. Pressure is \(101.3 \mathrm{kPa}\). The column has a 2.0 -in. head of liquid on...
-
Tretter alleged that his exposure over the years to asbestos products manufactured by Philip Carey Manufacturing Corporation caused him to contract asbestosis. Tretter brought an action against Rapid...
-
answer should not be copied from any websites. Two economists are attending a conference in an unfamiliar city. At the end of the day, Economist A states she is "in the mood for a high-quality...
-
A gaseous fuel with a volumetric analysis of 60 percent CH4, 30 percent H2, and 10 percent N2 is burned to completion with 130 percent theoretical air. Determine (a) The airfuel ratio and (b) The...
-
Complete hydrogenation of 1,3,5,7-cyclooctatetraene is exothermic by -101 kcal mol -1 . Hydrogenation of cyclooctene proceeds with H = 223 kcal mol -1 . Are these data consistent with the description...
-
Following are spectroscopic and other data for several compounds. Propose a structure for each of them. (a) Molecular formula = C 6 H 4 Br 2 . 1 H NMR spectrum A. 13 C NMR: 3 peaks. IR: v = 745 (s,...
-
Graph each inequality. y 4-2x
-
What are the functions of audit working papers?
-
Explain the different types of evaluation that must be done at the completion of an audit.
-
\(\alpha=0.025\), d.f. \(_{\mathrm{N}}=7\), d.f. \(_{\mathrm{D}}=3\) Find the critical F-value for a right-tailed test using the level of significance \(\alpha\) and degrees of freedom d.f.N and...
-
Since the Exxon Valdez incident brought surface contamination to the evening news, the press has been replete with references to environmental disasters, making most everyone conscious of the...
-
Outline a set of five guidelines for the preparation of working papers.
-
For tetragonal crystals, cite the indices of directions that are equivalent to each of the following directions: (a) [011] (b) [100]
-
Q1) What is the a3 Value Q2) What is the a7 Value Q3) What is the a4 Value Q4) What is the b3 Value Q5) What is the b2 Value Q6) What is the sign of 2nd constraint? A pastry chef at a bakery wants to...
-
The dipole moment of bromobenzene is 5. 17 X 10-30 C m and its polarizability volume is approximately 1.5 x 10-29 m3. Estimate its relative permittivity at 25C, when its density is 1491 kg m-3.
-
Calculate the vapour pressure of a spherical droplet of water of radius 20.0 nm at 35.0e. The vapour pressure of bulk water at that temperature is 5.623 kPa and its density is 994.0 kg m-1.
-
The contact angle for water on clean glass is close to zero. Calculate the surface tension of water at 30C given that at that temperature water climbs to a height of 9.11 cm in a clean glass...
-
1.When is the right time to use equivalent annual cost ( EAC) method ? and how we make the decision by using it. 2. Should financing costs be included as an incremental cash flow in capital budgeting...
-
ACCT 597 Assignment 5 The KJA partnership is formed by Kennedy, Jane, and Allyson. Kennedy will contribute $500,000 cash as will Jane. Allyson will contribute real property with a FMV of $800,000 and...
-
35. Devin and Merri are getting prequalified. Devin earns $1.300 a month and Merri earns $1,000 a month. Merri receives child support of $700 a month from an ex-husband. Devin gets $150 annual...

Study smarter with the SolutionInn App


