Write the expected major product of reaction of 1-propynyllithium, CH 3 C C - Li +
Question:
Write the expected major product of reaction of 1-propynyllithium, CH3C ≡ C-Li+, with each of the following molecules in THF.
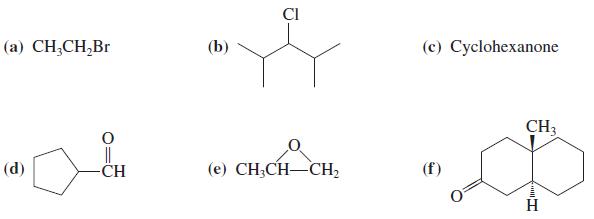
Transcribed Image Text:
CI (а) CН,CH,Br (b) (с) Суclohexanone CH3 (d) -СH (е) CН,CH—СH (f)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)

Answered By

Yuvaraj A R
My Qualification:
Post-Doctorate in Chemistry and Materials
PhD in Chemistry and Advance Material
MSc in Organic Chemistry
BSc in Chemistry, Mathematics and Physics
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the expected major product of each of the following reactions. (a) (b) (c) (d) CH CH,CH,OH COnC HI Conc. HBr (CH,)CHCH.CH.OH- Conc. HI OH CH,CH2,COH COn HCT
-
Write the expected major product of reaction of each of the carbonyl compounds (i) (iii) with each of the reagents (a) (h). CH2CH,CH3 (i) (ii) CH;CH=C. (iii) CH || () , P, ,C- (c) Cl2, CCl, (e)...
-
Give the expected major product of reaction of 2,2-dimethyloxacyclopropane with each of the following reagents? (a) Dilute H2SO4 in CH3OH (b) Na+ -OCH3 in CH3OH (c) Dilute, aqueous HBr (d)...
-
List and describe the payment options (terms of sale) that can be applied to domestic U.S. shipments.
-
Describe the Critical Chain method and compare it to CPM.
-
The Morocco Company uses a standard cost accounting system and estimates production for the year to be 60,000 units. At this volume, the company's variable overhead costs are $0.50 per direct labor...
-
The following information is taken from the annual report of The Mann Corporation: Calculate the receivable turnover ratio and the receivable collection period for 2014 and 2015. How much additional...
-
Based on the information below, record the adjusting journal entries that must be made for Garibaldi Consulting on June 30, 2016. The company has a June 30 fiscal year-end. Use 18 as the page number...
-
3. Read Management Decision Problem #2 on p. 362 and answer questions posed by the problem.
-
Research has found that the target market selected by the maker of a digital camera consists primarily of individuals who are other-directed and also have a high need for cognition. How can the...
-
(a) Write the expected product of the reaction of 3-octyne with Na in liquid NH 3 . (b) When the same reaction is carried out with cyclooctyne (Problem 33b), the product is cis-cyclooctene, not...
-
Write the mechanism and final product for the reaction of 1-propynyllithium with trans-2,3- dimethyloxacyclopropane.
-
In 2021, Meghann, a single taxpayer, has QBI of $110,000 and modified taxable income of $78,000 (this is also her taxable income before the QBI deduction). Given this information, what is Meghanns...
-
List the three methods outlined in Standard 560 that should be utilized in a quality assurance program for the internal auditing department. What is the controversy involving external reviews?
-
Discuss the relative costs of the two different audit strategies.
-
Who is responsible for coordinating internal and external auditing activities? Who bears the ultimate responsibility of all auditing activities? How does the director coordinate internal and external...
-
Explain the interrelationships among audit risk, materiality and audit evidence.
-
Identify the four phases of a financial statement audit and discuss the time frame in which each of these occurs.
-
For the pre-deposition heat treatment of a semiconducting device, gallium atoms are to be diffused into silicon at a temperature of 1150(C for 2.5 h. If the required concentration of Ga at a position...
-
Teasdale Inc. manufactures and sells commercial and residential security equipment. The comparative unclassified balance sheets for December 31, 2015 and 2014 are provided below. Selected missing...
-
Explicit expressions for hydrogenic orbitals are given in Tables 10.1 and 9.3. (a) Verify both that the 3px orbital is normalized (to I) and that 3px and 3dxy are mutually orthogonal. (b) Determine...
-
Show that l, and 12 both commute with the Hamiltonian for a hydrogen atom. What is the significance of this result?
-
Some atomic properties depend on the average value of 1/r rather than the average value of r itself. Evaluate the expectation value of 1/, for? (a) A hydrogen 15 orbital, (b) A hydrogenic 25 orbital,...
-
I'm comparing the age for educated Male-owned businesses and the age for educated Women-owned businesses by using the result below that I got from my data. * I want to analyze and summarize my data...
-
Hi i am using excel to calculate my NPV. when calculating NPV should i include the year zero in the calculation and if i should how should i calculate it?
-
1 . Assume today s settlement price on a CME EUR futures contract is . You have a short position in one contract. Your performance bond account currently has a balance of $ 1 , 7 0 0 . The next three...

Study smarter with the SolutionInn App


