A steel microstructure contains 92% martensite and 8% Fe 3 C; the composition of the martensite is
Question:
A steel microstructure contains 92% martensite and 8% Fe3C; the composition of the martensite is 1.10% C. Using Figure 12-35, determine
(a) The temperature from which the steel was quenched and
(b) The carbon content of the steel.
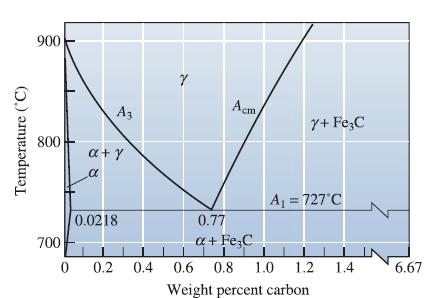
Transcribed Image Text:
Temperature (°C) 900 800 700 0 A3 a+y α 0.0218 0.2 0.4 Y I Acm Y+ Fe C A₁ = 727°C 0.77 a + Fe C Tī 0.6 0.8 1.0 1.2 1.4 Weight percent carbon 6.67
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
a The temperature 1250C so that the martensite is in ...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
A steel microstructure contains 75% marten site and 25% ferrite; the composition of the marten site is 0.6% C. Using Figure 12-32, determine (a) The temperature from which the steel was quenched and...
-
The composition of timothy hay is 11.3% protein, 1.6% fat and 41% crude fiber (as-fed basis). How many pounds of protein, fat, and crude fiber are present in a ton of hay?
-
The composition of moist air is given on a molar basis to be 78 percent N2, 20 percent O2, and 2 percent water vapor. Determine the mass fractions of the constituents of air.
-
You are in the market for a new car and have narrowed your search down to two types: Car X costs $36,300, will last for four years, and will be worth $3,630 at the end of its useful life. It will...
-
Without looking at the text, derive a nations offer curve from its production frontier, its indifference map, and two relative commodity prices at which trade could take place (i.e., sketch a figure...
-
Silicon carbide (SiC) has a specific gravity of 3.1 g/cm3. A sintered SiC part is produced, occupying a volume of 500 cm3 and weighing 1200 g. After soaking in water, the part weighs 1250 g....
-
Write up a two-column cash book from the following: 19X9 Nov 1 Balance brought forward from last month: Cash 105; Bank 2,164. 2 Cash sales 605. 55 3 55 35 35 23 35 2 25 " " " " 4 5 6 7 Took 500 out...
-
The Kelly Services, Inc., and Subsidiaries balance sheets from its 2008 annual report are presented in Exhibit 5-4. Required a. Using the balance sheets, prepare a vertical common-size analysis for...
-
Your company is evaluating a new project. It expects to sell 5,500 units per year at $69 net cash flow apiece for the next 10 years. In other words, the annual operating cash flow is projected to be...
-
A binary mixture of mole fraction zj is flashed (o conditions T and P, Fur one of the following determine: the equilibrium mole fractions x1 and y1 of the liquid and vapor phases formed, the molar...
-
What step controls the rate of martensitic phase transformations?
-
What is the difference between isothermal and athermal transformations?
-
Determine an expansion for the heat transfer in a closed system isochoric process.
-
How many real positive values of \(r\) satisfy the following. (a) \(T(r, 0.1)=1\) (b) \(T(r, 0.5)=0.5\) (c) \(T(r, 0)=3\)
-
A machine is subject to a harmonic excitation with an amplitude of \(15,000 \mathrm{~N}\). The force transmitted to the floor through an isolator has an amplitude of \(3000 \mathrm{~N}\). What...
-
Convert \(1000 \mathrm{rpm}\) to \(\mathrm{rad} / \mathrm{s}\).
-
A \(50 \mathrm{~kg}\) machine is mounted on an isolator with a stiffness of \(6 \times 10^{5} \mathrm{~N} / \mathrm{m}\). During operation, the machine is subject to a harmonic excitation with a...
-
A machine operates at \(100 \mathrm{rad} / \mathrm{s}\) and has a rotating component of mass \(5 \mathrm{~kg}\) whose center of mass is \(3 \mathrm{~cm}\) from the axis of rotation. What is the...
-
The article "Supervised Exercise Versus Non- Supervised Exercise for Reducing Weight in Obese Adults" (The J. of Sports Med. and Physical Fitness, 2009: 85-90) reported on an investigation in which...
-
Q1) What is the a3 Value Q2) What is the a7 Value Q3) What is the a4 Value Q4) What is the b3 Value Q5) What is the b2 Value Q6) What is the sign of 2nd constraint? A pastry chef at a bakery wants to...
-
An automobile assembly plant produces 400 vehicles per day. Make an order-of-magnitude estimate for the weight of the steel needed to make those vehicles. Explain and justify the reasonableness of...
-
Think of some quantity that you encounter in your day-to-day life for which it would be difficult to obtain a highly accurate numerical value but for which an order-of-magnitude approximation can be...
-
The modulus of elasticity, modulus of rigidity, Poissons ratio, and the unit weight for various materials are shown below. The data is given as Material; Modulus of Elasticity, E (Mpsi & GPa);...
-
. The Sarbanes-Oxley Act of 2002 requires management to offer an opinion on the fairness of the financial statements each year. A True B False. Question10 ( 5Points 3 Pull manufacturing always...
-
Wolfpack Company is a merchandising company that is preparing a budget for the month of July. It has provided the following information: Wolfpack Company Balance Sheet June 30 09 Assets Cash Accounts...
-
The condensed income statement for Hayden Corp. for the past year is as follows: Product T Product U Sales $680,000 $320,000 Costs: Variable costs $(540,000) $(220,000) Fixed costs Total costs Income...

Study smarter with the SolutionInn App


