Consider the following mixture of SO 2 (g) and O 2 (g). If SO 2 (g) and
Question:
Consider the following mixture of SO2(g) and O2(g).
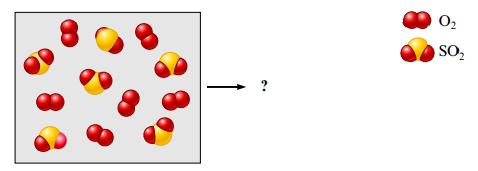
If SO2(g) and O2(g) react to form SO3(g), draw a representation of the product mixture assuming the reaction goes to completion. What is the limiting reactant in the reaction? If 96.0 g of SO2 react with 32.0 g O2, what mass of product will form?
Transcribed Image Text:
? 0₂ SO₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
a The limiting reactan...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
What is a limiting reactant in a reaction mixture? Explain how it determines the amount of product.
-
What is a limiting reactant problem? Explain the method you are going to use to solve limiting reactant problems.
-
Describe how you could separate the following mixture of metal ions: Cd2+, Pb2+, and Sr2+.
-
The number x of bicycle helmets people are willing to buy per week from a retail chain at a price of $p is given by x = 1,000 - 60p + 25 20 ¤ p ¤ 100 (see the figure). (A) Find dx/dp....
-
Because expenses incurred both in a business and for the production of investment income are deductible, why is it important to determine in which category a particular activity falls?
-
The reaction SO2 1g2 + 2 H2S(g) 3 S(s) + 2 H2O (g) is the basis of a suggested method for removal of SO2 from power-plant stack gases. The standard free energy of each substance is given in Appendix...
-
8. Lloyd Dace worked for ACF Industries as a supervisor in the punchpress department of a carburetor factory. ACF demoted Dace to an hourly job on the assembly line, and
-
Backwoods American, Inc., produces expensive water repellent, down-lined parkas. The company implemented a total quality-management program in 2005. Following are quality-related accounting data that...
-
White Waters Amusement Parks currently pays a $4.10 common stock dividends. Dividends have been recently growing at a 14 percent annual rate and are expected to continue growing at this rate for the...
-
A firm sells five types of womens parkas. Some data follow. Parka Price Cost Salvage NV Q NV EP A $220 167.2 132 1,100 300 1,176 $47,949 B 205 155.8 127.1 2,000 800 2,269 74,992 C 190 144.4 121.6...
-
You know that chemical A reacts with chemical B. You react 10.0 g A with 10.0 g B. What information do you need to determine the amount of product that will be produced? Explain.
-
Consider an iron bar on a balance as shown. As the iron bar rusts, which of the following is true? Explain your answer. a. The balance will read less than 75.0 g. b. The balance will read 75.0 g. c....
-
Describe the changing status of HRM. What factors have led to these changes?
-
You require 3.200 kg of chicken meat to produce a farce. A chicken size 16 has a net yield of 70 % meat. a.What is the total weight of chicken you would require expressed in 0.000 Kg? (round your...
-
Jannette purchased $550 worth of inventory from Jack on credit. Jacks cost of good was $100. What is the journal entry for Jack?
-
The human service model is concerned with the interaction between the individual and the environment, stressing the need for balance between the two. What are the characteristics of the human...
-
While running the PCC, what other strategic areas should you be working on?
-
Assuming CAPM holds, we have: Stock A: E(r) = 9%, Standard Deviation = 10% Stock B: E(r) = 4%, SD = 8% Stock C: E(r) = 11%, SD = 14% Market: E(r) = 9%, SD = 10% T-Bills: E(r) = 4%, SD = 0 Which has...
-
Three fire hoses are connected to a fire hydrant. Each hose has a radius of 0.020 m. Water enters the hydrant through an underground pipe of radius 0.080 m. In this pipe the water has a speed of 3.0...
-
The column shown in the figure is fixed at the base and free at the upper end. A compressive load P acts at the top of the column with an eccentricity e from the axis of the column. Beginning with...
-
Prove that the perfect gas temperature scale and the thermodynamic temperature scale based on the Second Law of thermodynamics differ from each other by at most a constant numerical factor.
-
Evaluate (ClS/ClV)]' for (a) A van der Waals gas, (b) A Dieterici gas (Table 1.7). For an isothermal expansion, for which kind of gas (and a perfect gas) will /).5be greatest? Explain your conclusion.
-
Two of the four Maxwell relations were derived in the text, but two were not. Complete their derivation by showing that (S/V)T = (p/T)V (T/P)s = (V/S)p
-
"Ecommerce is the newest and most disruptive distribution channel wherein 1 po goods and services are represented virtually online and then distributed directly to the buyer" Agree Disagree
-
1) What is the system and service manager on our Fedora VMs?How can you verify that on your system? (show the command below).What version of Linux is running on fawad, and what is it's system...
-
7)The terminal value is always "placed" in the last year of a DCF forecast. If the terminal value is placed in year 2024 (as it is in Case 2, for P&G), this value represents...

Study smarter with the SolutionInn App


