Esterification reactions are carried out in the presence of a strong acid such as H 2 SO
Question:
Esterification reactions are carried out in the presence of a strong acid such as H2SO4. A carboxylic acid is warmed with an alcohol, and an ester and water are formed. You may have made a fruity-smelling ester in the lab when studying organic functional groups. Name the carboxylic acid that is necessary to complete the following esterification reaction.
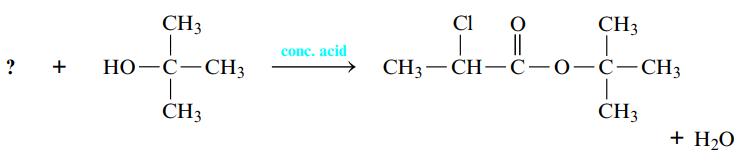
Transcribed Image Text:
? + CH3 | HO-C-CH3 T CH3 conc. acid Cl O 98 || CH3—CH—C—0—C—CH3 CH3 1 I CH3 + H₂O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
The carboxylic acid that is necessary is Acetic Acid Esterifi...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
In the presence of a platinum catalyst, ammonia, NH3, burns in oxygen, O2, to give nitric oxide, NO, and water vapor. How many volumes of nitric oxide are obtained from one volume of ammonia,...
-
When ethyl 4-hydroxybutyrate is heated in the presence of a trace of a basic catalyst (sodium acetate), one of the products is a lactone. Propose a mechanism for formation of this lactone.
-
In the presence of a tungsten catalyst at high temperatures, the decomposition of ammonia to nitrogen and hydrogen is a zero-order process. If the rate constant at a particular temperature is 3.7 ...
-
20 -101 10 in- laminate substrate Fig.2 Q2: The tool shown in Fig.2 is used in a gluing operation to press a thin laminate to a thicker substrate. If the wheels at points A and B both have 2 in...
-
The Triton Energy Corporation explores for and produces oil and gas. Company president Gail Freeman wants to have her company's analyst forecast the company's sales per share for 2000. This will be...
-
In Exercises 3-6, solve the initial value problem x'(t) = Ax(t) for t > 0, with x(0) = (3, 2). Classify the nature of the origin as an attractor, repeller, or saddle point of the dynamical system...
-
What is an open-end mutual fund?
-
Cortez Company sells chairs that are used at computer stations. Its beginning inventory of chairs was 100 units at $60 per unit. During the year, Cortez made two batch purchases of this chair. The...
-
After a thorough investigation of the major differences between internal and external auditing explain five ways on how the auditor general could be both external and internal auditor in his public...
-
Go to the Structural Styles section and create a style rule that sets the background color of the browser window to rgb(173, 189, 227). Create a style rule for the body element that sets the...
-
An organometallic compound is one containing at least one metalcarbon bond. An example of an organometallic species is (CH 3 CH 2 )MBr, which contains a metalethyl bond. a. If M 2+ has the electron...
-
Ignoring ring compounds, which isomer of C 2 H 4 O 2 should boil at the lowest temperature?
-
You run a die casting facility that produces metal tools used in industry. (Dies are essentially molds that are filled with superhot molten metal to make metal products.) Competition is fierce in...
-
Briefly explain two major concerns that we should have as auditors about the way information systems jobs are defined.
-
Anyone working for a company, a project, a joint venture, or in a group thats been together even for a few months is working in a cultural system. Human beings cant work around each other for very...
-
Briefly explain the difference between policies and standards. What are top management's responsibilities with respect to information systems policies and standards?
-
Mark, a project leader in Austin, Texas, needed a new software engineer for his eight-person team. He used his network, reviewed rsums, and invited 15 candidates for interviews. In addition he had...
-
Chairs? There is not much that can be said about chairs, right? Some are comfortable, some are not; some are cushy, some are firm. Since that is just about the whole story on the subject, I never...
-
Use the following India balance of payments data from the IMF (all items are for the current account) to answer questions 3.5 through 3.9. 1. What is India's balance on goods? 2. What is India's...
-
Why do CPA firms sometimes use a combination of positive and negative confirmations on the same audit?
-
Write the structure of a representative segment of polyurethane prepared by reaction of ethylene glycol with MDI.
-
The smoking salons of the Hindenburg and other hydrogen-filled dirigibles of the 1930s were insulated with urea formaldehyde polymer foams. The structure of this polymer is highly cross-linked, like...
-
The polymeric resin used for Merrifield solid-phase peptide synthesis (Section 26.8) is prepared by treating polystyrene with N-(hydroxymethyl) phthalimide and trifluoromethanesulfonic acid, followed...
-
Tyler believes that some laws passed by his state's legislature are bad, and he refuses to comply with them. He believes a universal law above all others that grants rights to all people and that it...
-
Organizational commitment increases the likelihood that an individual will respond to a negative work event with loyalty or neglect. voice or exit. loyalty or voice. O exit or loyalty.
-
In the context of decision-making, satisficing refers to Selecting options at random without any thought O Always choosing the first available option O Avoiding decisions to reduce cognitive load...

Study smarter with the SolutionInn App


