Air enters an adiabatic nozzle steadily at 300 kPa, 200C, and 45 m/s and leaves at 100
Question:
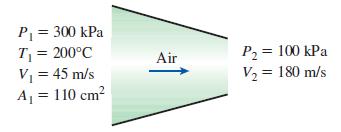
Air enters an adiabatic nozzle steadily at 300 kPa, 200°C, and 45 m/s and leaves at 100 kPa and 180 m/s. The inlet area of the nozzle is 110 cm2. Determine
(a) The mass flow rate through the nozzle
(b) The exit temperature of the air
(c) The exit area of the nozzle.
Transcribed Image Text:
P= 300 kPa T = 200°C V = 45 m/s A = 110 cm? P2 = 100 kPa V2 = 180 m/s Air %3D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
Air is accelerated in a nozzle from 45 ms to 180 ms The mass flow rate the exit temperature and the ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
Air enters an adiabatic nozzle steadily at 300 kPa, 200°C, and 30 m/s and leaves at 100 kPa and 180 m/s. The inlet area of the nozzle is 80 cm2. Determine (a) The mass flow rate through the...
-
Reconsider Prob. 539. Data From Q#39: Air enters an adiabatic nozzle steadily at 300 kPa, 200C, and 45 m/s and leaves at 100 kPa and 180 m/s. The inlet area of the nozzle is 110 cm 2 . Determine (a)...
-
Air enters an adiabatic nozzle steadily at 400kPa, 200oC and 35m/s and leaves at 150kPa and 180 m/s. The inlet area of the nozzle is 75cm2. Determine (a) The mass flow rate. (b) The exit temperature...
-
Allison is paid $1,520 per week. What is the amount of federal income tax withheld from Allisons paycheck under the following conditions? Use the percentage method table in the Appendix to this...
-
The following data represent the number of days absent per year in a population of six employees of a small company: 1 3 6 7 9 10 a. Assuming that you sample without replacement, select all possible...
-
Propose structures for the three compounds whose 1H NMR spectra are shown. (a) C 5 H 10 O (b) C 7 H 7 Br (c) C 8 H 9 Br TMS O ppm 10 Chemical shift (8) TMS O ppm 10 9. 8. 3 2 Chemical shift (8) TMS O...
-
8. Question: DatagraphiX manufactured and sold computer graphics equipment that allowed users to transfer large volumes of information directly from computers to microfilm. Customers were required to...
-
Your boss at Scott Sampson Products, Inc., has just provided you with the schedule and lead times for the bracket in Problem 14.8. The unit is to be prepared in week 10. The lead times for the...
-
Current Attempt in Progress Kennel Corporation, a publicly traded company, was organized on January 1, 2021. It is authorized to issue an unlimited number of $3 noncumulative preferred shares and an...
-
With the growth in demand for exotic foods, Possum Products's CEO Michael Munger is considering expanding the geographic footprint of its line of dried and smoked low-fat opossum, ostrich, and...
-
Consider an adiabatic turbine operating steadily. Does the work output of the turbine have to be equal to the decrease in the energy of the steam flowing through it?
-
A house is maintained at 1 atm and 24C, and warm air inside a house is forced to leave the house at a rate of 90 m 3 /h as a result of outdoor air at 5C infiltrating into the house through the...
-
The following information was taken from the books of Tapwater Company. Compute the amount of net cash provided by (used in) operating activities during 2011 using the indirectmethod. Dec. 31, 2011...
-
Jessica owns 250 acres of rental property. Jessica gives Melissa the right to any rental income that the property earns in the next eight years. What if Jessica tells Melissa that she can keep 50...
-
Explain Python programming for numerical computation.
-
YesChairs Inc. produces 3 types of chairs for 3 sizes of buyers. Overhead costs incurred during the manufacturing process include: Machine maintenance bill Quality control bill SS $ $ 85,000 65,000...
-
Translate the following code into MIPS code. int Sum (int a) { } if (a <3) return (1); else return (a + Sum (a - 2) + Sum (a-3));
-
Presented below is information related to Ivanhoe Co., owned by D. Martin, for the month of January 2024. Ending inventory per perpetual records $21,300 Rent expense $21,200 Ending inventory actually...
-
If you borrow $800 for 6 months at 16% annual simple interest, how much must you repay at the end of the 6 months?
-
A company pledges their receivables so they may Multiple Choice Charge a factoring fee. Increase sales. Recognize a sale. Collect a pledge fee. Borrow money. Failure by a promissory notes' maker to...
-
Determine the specific volume of superheated water vapor at 3.5 MPa and 4508C based on (a) The ideal-gas equation, (b) The generalized compressibility chart, and (c) The steam tables. Determine the...
-
Somebody claims that oxygen gas at 160 K and 3 MPa can be treated as an ideal gas with an error of less than 10 percent. Is this claim valid?
-
Ethane in a rigid vessel is to be heated from 50 psia and 1008F until its temperature is 540oF. What is the final pressure of the ethane as predicted by the compressibility chart?
-
. Exercise 9-33 and 9-39 {Algoi Upriver Parts manufactures two prod ucts. v-1 and v-2. at its River Plant. Selected data for an average month for the two products follow. Units produced Direct...
-
Problem 08-11 (Static) [LO 8-2] In year 1, Maxim sold investment land with a tax basis of $77,000. Payment consisted of $10,000 cash down and the purchaser's note for $90,000. The note is payable in...
-
Payback Period : Determine the payback period for an investment with an initial cost of $200,000 and annual cash inflows of $50,000. How long will it take for the company to recover its investment?

Study smarter with the SolutionInn App


