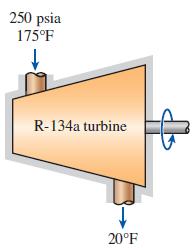
R-134a vapor enters into a turbine at 250 psia and 175F. The temperature of R-134a is reduced
Question:
R-134a vapor enters into a turbine at 250 psia and 175°F. The temperature of R-134a is reduced to 20°F in this turbine while its specific entropy remains constant. Determine the change in the enthalpy of R-134a as it passes through the turbine.
Transcribed Image Text:
250 psia 175°F R-134a turbine 20°F
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
R134a is explained in a turbine during which the ent...View the full answer

Answered By

Pharashram rai
i am highly creative, resourceful and dedicated freelancer an excellent record of successful classroom presentations and writing . I have more than 4 years experience in tutoring students especially by using my note making strategies and engineering field . Especially adept at teaching methods of maths and writing , and flexible teaching style with the willingness to work beyond the call of duty. Committed to ongoing professional development and spreading the knowledge within myself to the blooming ones to make them fly with .
4.80+
65+ Reviews
270+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
In the ideal Rankine power cycle with reheat of Fig. 8.33, steam leaves the steam generator at 1200 psia and 1200 8 F with a mass flow rate of 50 lbm/s. It leaves the high-pressure turbine at 150...
-
The stators in a gas turbine are designed to increase the kinetic energy of the gas passing through them adiabatically. Air enters a set of these nozzles at 300 psia and 700oF with a velocity of 80...
-
A flow calorimeter is a device used to measure the specific heat of a liquid. Energy is added as heat at a known rate to a stream of the liquid as it passes through the calorimeter at a known rate....
-
Shown here is a list published by Electronics Weekly.com of the top five semiconductor companies in the United States by revenue ($ billions). a. Construct a bar chart to display these data. b....
-
A non-dividend-paying common stock is trading at $100. Suppose you are considering a European put option with a strike price of $110 and one year to expiration. What is the annually compounded...
-
Calculating Flotation Costs the Educated Horses Corporation needs to raise $40 million to finance its expansion into new markets. The company will sell new shares of equity via a general cash...
-
What are three pieces of information about corporate bonds that you could find in the bond section of a newspaper?
-
Bad-Debt Reporting Presented below is information related to the Accounts Receivable accounts of Gulistan Inc. during the current year 2010. 1. An aging schedule of the accounts receivable as of...
-
Financial analyst forecast XYZ company's growth for the future to be constant 8%. XYZ recent dividend was $0.88. What is the value of XYZ stock when the required return is 12%?
-
Sullivans Island Company began operating a subsidiary in a foreign country on January 1, 2020, by investing capital in the amount of 60,000 pounds. The subsidiary immediately borrowed 140,000 pounds...
-
Using the relation ds = (Q/T) int rev for the definition of entropy, calculate the change in the specific entropy of R-134a as it is heated at a constant pressure of 200 kPa from a saturated liquid...
-
Water vapor enters a compressor at 35 kPa and 160C and leaves at 300 kPa with the same specific entropy as at the inlet. What are the temperature and the specific enthalpy of water at the compressor...
-
Explain the operation of a demand controlled ventilation (DCV) system.
-
Define refrigeration and air conditioning.
-
The team is in the process of completing the work breakdown structure of the project. Once they have completed this, which of the following can they begin to work on? a. Estimating costs b....
-
Payroll is often used as a good example of batch processing using sequential files. Explain why.
-
Define internal energy and enthalpy.
-
The project manager always involves the team in the creation of the work breakdown structure. What is the most significant benefit derived from this approach? a. Generation of a more accurate...
-
The Ace Jewelry Store sold a necklace for $144. If the percent profit (based on cost) equals the cost of the necklace to the store, how much did the store pay for it? Use where P is profit and C is...
-
On August 31, 2012, the balances of the accounts appearing in the ledger of Wood Interiors Company, a furniture wholesaler, are as follows:Prepare the August 31, 2012, closing entries for Wood...
-
A Carnot heat engine receives heat from a reservoir at 1700F at a rate of 700 Btu/min and rejects the waste heat to the ambient air at 80F. The entire work output of the heat engine is used to drive...
-
The structure of a house is such that it loses heat at a rate of 3800 kJ/h per C difference between the indoors and outdoors. A heat pump that requires a power input of 4 kW is used to maintain this...
-
An air-conditioner with refrigerant-134a as the working fluid is used to keep a room at 23°C by rejecting the waste heat to the outdoor air at 34°C. The room gains heat through the walls and...
-
#3. The following table depicts current market conditions (assume annual compounding): < ABC Year Current spot rates (r.) < Implied 2-year forward rate (f2,+) < 4 2.30% 5 2.40% 60 2.45% 7 < 2.60% 80...
-
The fixed - cost component of transportation refers to costs which change with the volume of material moved. True False
-
If a VC crafted its offer using the Venture Capital Method of valuation which means: A.None of the provided options. B.It prefered a DCF valuation instead of a comparable company valuation. C.It used...

Study smarter with the SolutionInn App


