Initially 10 kg of saturated liquid water at 400 K and 2 kg of saturated water vapor
Question:
Initially 10 kg of saturated liquid water at 400 K and 2 kg of saturated water vapor at 300 K are together in a sealed, constant volume adiabatic container. After a time the water adjusts to a uniform state within the tank. What are the pressure and temperature in this final state, and what are the final volumes of the liquid and vapor? The graphs in Appendix A.3 may be used along with the tables in solving this problem.
Data From Appendix A.3
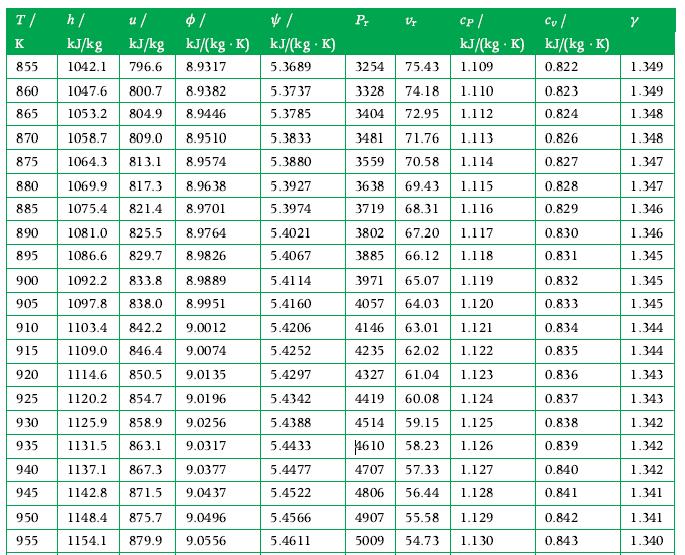
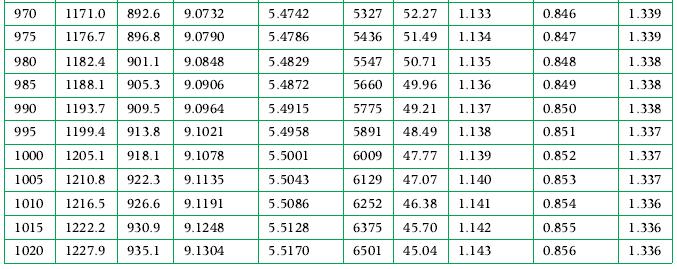
Transcribed Image Text:
T/ h/ K 855 860 865 870 875 u/ kJ/kg kJ/kg 1042.1 796.6 8.9317 1047.6 800.7 8.9382 1053.2 804.9 8.9446 8.9510 813.1 8.9574 1058.7 809.0 1064.3 880 1069.9 817.3 8.9638 885 1075.4 821.4 8.9701 890 1081.0 825.5 8.9764 895 1086.6 829.7 8.9826 900 1092.2 833.8 905 1097.8 838.0 910 915 920 925 930 935 $/ kJ/(kg.K) 8.9889 8.9951 1103.4 842.2 9.0012 1109.0 846.4 9.0074 1114.6 850.5 9.0135 1120.2 854.7 9.0196 1125.9 858.9 9.0256 1131.5 863.1 9.0317 9.0377 9.0437 940 1137.1 867.3 945 1142.8 871.5 950 1148.4 875.7 955 1154.1 879.9 9.0496 9.0556 4/ kJ/(kg. K) 5.3689 5.3737 5.3785 5.3833 5.3880 5.3927 5.3974 5.4021 5.4067 5.4114 5.4160 5.4206 5.4252 5.4297 5.4342 5.4388 5.4433 5.4477 5.4522 5.4566 5.4611 Pr Vr Cp / Cu / kJ/(kg. K) kJ/(kg .K) 1.109 0.822 3254 75.43 3328 74.18 1.110 3404 72.95 1.112 3481 71.76 1.113 3559 70.58 1.114 3638 69.43 1.115 3719 68.31 1.116 3802 67.20 1.117 3885 66.12 1.118 3971 65.07 4057 64.03 4146 63.01 1.121 4235 62.02 1.122 4327 61.04 1.123 4419 60.08 1.124 4514 59.15 1.125 4610 58.23 1.126 4707 57.33 1.127 4806 56.44 1.128 4907 55.58 1.129 5009 54.73 1.130 1.119 1.120 0.823 0.824 0.826 0.827 0.828 0.829 0.830 0.831 0.832 0.833 0.834 0.835 0.836 0.837 0.838 0.839 0.840 0.841 0.842 0.843 Y 1.349 1.349 1.348 1.348 1.347 1.347 1.346 1.346 1.345 1.345 1.345 1.344 1.344 1.343 1.343 1.342 1.342 1.342 1.341 1.341 1.340
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (7 reviews)
To solve this problem we can apply the principle of energy equilibrium and the ideal gas law Here ar...View the full answer

Answered By

Anik Kumar Bosu
I am expert in Maths, Physics, Chemistry and Biology. I also teach student from 3 years. Students becomes satisfied by my teaching. So, do not worry.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Fundamentals And Engineering Applications
ISBN: 9780521862738
1st Edition
Authors: William C. Reynolds, Piero Colonna
Question Posted:
Students also viewed these Engineering questions
-
A well-insulated rigid vessel contains 3 kg of saturated liquid water at 40oC. The vessel also contains an electrical resistor that draws 10 amperes when 50 volts are applied. Determine the final...
-
The 4 kg of saturated liquid water in Fig. 2.30 are completely vaporized at a constant pressure of 200 kPa. Determine the volume of the water at state 1 and at state 2. Figure 2.30 State 1 State 2...
-
A rigid tank initially contains 1.4-kg saturated liquid water at 200oC. At this state, 25 percent of the volume is occupied by water and the rest by air. Now heat is supplied to the water until the...
-
Measuring the height of a California redwood tree is very difficult because these trees grow to heights over 300 feet. People familiar with these trees understand that the height of a California...
-
Record the following events of Mayo Health Clinic, a VHWO: 1. In her will, a leading citizen left a bequest of $400,000 to the clinic. Stipulations were that the amount was to become the corpus of a...
-
Civil Code 1719, subdivision (a) provides in part that any person who draws a check that is dishonored due to insufficient funds shall be liable to the payee for the amount owing upon the check and...
-
Why is the fact that moneylenders marginal costs are below average costs a hallmark of monopolistic competition?
-
Wagner Inc. is a large Canadian public company that uses IFRS. A lease for a fleet of trucks has been capitalized and the lease amortization schedule for the first three lease payments appears below....
-
Question 11 (3 points) Find the following Probability: P(not a 5) '
-
A one-dimensional harmonic oscillator has an infinite series of equally spaced energy states, with E x = sh, where s is a positive integer or zero, and is the classical frequency of the oscillator....
-
The gas in a pistoncylinder system undergoes a change of U = 30 kJ during a process in which 40 kJ of energy as work are transferred from the gas to the piston. Was there energy transfer as heat...
-
Liquid oxygen (commonly referred to as LOX) in a rocket-propellant tank is kept at a pressure of approximately 1 bar. A certain ullage volume is occupied by saturated vapor for safety. Use the graph...
-
On the moon, the gravitational acceleration is approximately one-sixth that on the surface of the earth. A 5-kg mass is weighed with a beam balance on the surface of the moon. What is the expected...
-
Albert purchased a tract of land for $ 1 4 0 , 0 0 0 in 2 0 2 0 when he heard that a new highway was going to be constructed through the property and that the land would soon be worth $ 2 0 0 , 0 0 0...
-
1. What procedure was performed? 2. Search the index for the main terms/sub terms and record the selections? 3. What code(s) does the index guide you to review? 4. What CPT code and Modifier should...
-
You are an HR manager at that company and hiring for the position you had. What job are you hiring for (your previous or current position)? List some details. Provide basic background information...
-
Explain why career development is value to organization? Also explain the 5 traditional stages involved in a career with suitable examples.
-
Discuss two of the social media trends mentioned in the article that you find the most impactful. Give an examples of each trend....
-
The inductors in Fig. 6.87 are initially charged and are connected to the black box at t = 0. If i1(0) = 4 A, i2(0) = -2 A, and v(t) = 50e-200t mV, t ¥ 0$, find: (a) The energy initially stored...
-
Compile data on consumption and expenditures for the following categories in 30 different countries: (1) food and beverages, (2) clothing and footwear, (3) housing and home operations, (4) household...
-
Refer to the exercise of Problem P4.21. (a) Measure the angle of force A directly from the diagram and use it to find the magnitude of the force at hinge B. (b) A design condition is that the force...
-
A pair of large hydraulically operated shears in Figure P4.23 is attached to the end of the boom on an excavator. The shear is used for cutting steel pipe and I-beams during demolition work....
-
A cross section of the original design for the double-decker skyways in the Kansas City Hyatt Regency hotel is shown, along with the forces acting on the nuts and washers that support the upper and...
-
J Saved Hillyard Company, an office supplies specialty store, prepares its master budget on a quarterly basis. The following data have been assembled to assist in preparing the master budget for the...
-
Pine Corporation has sales revenue of $530,000 resulting in net operating income of $73,140. Average invested assets total $688,000. Residual income is $27,000. Calculate the new residual income if...
-
Your Corporation started a consulting business on January 1, 2019 by issuing $7,000 of common stock. What was net cash flow from operating activities? Provided services on account, $25,000. Incurred...

Study smarter with the SolutionInn App


