Suggest reagents that could be used to prepare these alkyl halides from alcohols: CH3 CH, b) CH,CI
Question:
Suggest reagents that could be used to prepare these alkyl halides from alcohols:
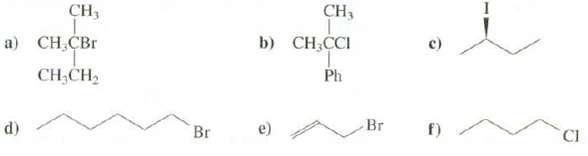
Transcribed Image Text:
CH3 CH, b) CH,CI а) CН,СВr Ph CH;CH2 Br CI Br г) d)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
CH3 a CH3COH CH3CH ...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Suggest a method that could be used to prepare this amine from an alkyl halide: PhCH2CH2NH2
-
Suggest indicators that could be used to assess an improvement in efficiency in each of the following areas: a. Reduce the energy used in production of goods and services b. Reduce the material...
-
Suggest an indicator that could be used to provide an end point for the titration of the first proton in H3AsO4.
-
ECB Co. has 1.2 million shares outstanding selling at $24 per share. It plans to repurchase 97,000 shares at the market price. What will be its market capitalization after the repurchase? What will...
-
After a more sophisticated analysis of Greenscape's costs, Drake has access to the following additional data. Required: Refer to the data in the previous case, as well as the additional data above....
-
A firm offers two products for sale. The marginal cost of one product is new zero once the first unit has been produced. The marginal cost of the other product rises as output rises. What would be...
-
Briefly define the following terms related to a note payable: present value of the note and maturity value of the note.
-
Data collected on the yearly demand for 50-pound bags of fertilizer at Wallace Garden Supply are shown in the following table. Develop a 3-year moving average to forecast sales. Then estimate demand...
-
At the beginning of the year, the AAA of Mauer, Inc., an S corporation, has a balance of $515,000. During the year, the following items occur. Operating income - $414,000 Interest income - 5,300...
-
Bonds 1. Municipal Bonds - Municipal bonds are haircut per Exhibit 1 based on both their time to maturity and scheduled maturity at date of issue. 2. Corporate Bonds - Corporate bonds are haircut...
-
Show all the steps in the mechanism for the reactions of 2-methyl-2-butanol with HCl in water.
-
Show the products of these reactions: CH,OH a) (CH,CH),NH + CH,CH,Br ether b) CH;CH,NCH3 + CH,I CH3
-
Assume that all the given functions have continuous second-order partial derivatives. If z = f(x, y), where x = r cos 0 and y = r sin 0, find (a) dz/or, (b) dz/ae, and (c) az/dr d0.
-
Last year, Ann invested her money in two purchases. She purchased a certificate of deposit for $1000 that paid 2% interest per year and purchased $3000 in corporate bonds paying 7% interest per year....
-
1. Suppose that you have two wavefunctions: 3/2 where do = 2 = YI V = (1) VII (1) 3/2 52.9 pm, p = (Z/ao)r. sin pe A cos (3 - p) pe (5 cos 0 1) sin 0e-i a. Show that I and II are orthogonal. Useful...
-
simplify the expression 1 0 8 x 8 2 + 4 8 x 8 2
-
Slove for x 6 ( x - 5 ) + 2 = 1 + 3 ( x - 3 )
-
Simplify 45 For 2/3, type in 2*sqrt(3) Question Help: Message instructor Calculator Submit Question
-
The directions of Coriolis component of acceleration, \(2 \omega V\), of the slider \(A\) with respect to the coincident point \(B\) is shown in Fig. 15. (1 to 4). Directions shown by figures (a) 2...
-
Below is a sample of the data in the file NFLAttendance which contains the 32 teams in the National Football League, their conference affiliation, their division, and their average home attendance....
-
Find each percent change. Round to nearest tenth of a percent. State if it is an increase or a decrease. From 42 to 41
-
Which substance in each of the following pairs is more reactive as a nucleophile? Explain. (a) (CH 3 ) 2 N or (CH 3 ) 2 NH (b) (CH 3 ) 3 B or (CH 3 ) 3 N (c) H 2 O or H 2 S
-
Rank the following compounds in order of their expected reactivity toward S N 2 reaction: CH 3 Br, CH 3 OTos, (CH 3 ) 3 CCl, (CH 3 ) 2 CHCl
-
Organic solvents such as benzene, ether, and chloroform are neither protic nor strongly polar. What effect would you expect these solvents to have on the reactivity of a nucleophile in SN2 reactions?
-
f If (t) = {2 (2,0
-
The spread of tobacco farming in seventeenth-century Virginia O a. helped to create a highly unequal society, dominated economically and politically by an elite plantation-owning class. O b....
-
Which one of the following statements accurately characterizes the British approach to governing the American colonies prior to 1763? O a. The British only occasionally intervened in the internal...

Study smarter with the SolutionInn App


