Suggest syntheses of these amino acids from the indicated starting materials: NH, Br a) PHCH,CHCO,H from PHCH,CHCO,H
Question:
Suggest syntheses of these amino acids from the indicated starting materials:
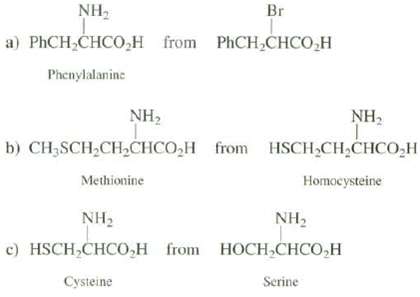
Transcribed Image Text:
NH, Br a) PHCH,CHCO,H from PHCH,CHCO,H Phenylalanine NH2 NH, b) CH3SCH2CHCHCO,H from HSCH CH,CHCO,H Methionine Homocysteine NH2 с) HSCH,CHCO,Н гom NH2 HОСH,CНCO,H Cysteine Serine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
Br a PhCHCHCOH excess NH3 NH ...View the full answer

Answered By

Marcus Solomon
I am committed to ensuring that my services always meet the clients' expectations.
4.60+
82+ Reviews
117+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show syntheses of these compounds from the indicated starting materials. More than one step may b necessary. Your syntheses may produce both enantiomers of any target that ischiral. , CH,CH,CH3 a)...
-
Propose syntheses of the following molecules from the indicated starting materials. Make use of any other reagents or solvents that you need. In some cases, there may be no alternative but to employ...
-
Show how Tagamet could be synthesized from the indicated starting materials. CH2OH CH CH SCH2CH2NH NHCH N CH CHS SCH CH3 +CH:NH2
-
Suggest two strategies for the company to minimise the impact of the strike on business operation?
-
In your own words, explain the difference between cost of goods manufactured and cost of goods sold.
-
Suppose that AT&T used benchmarking to compare the activity-based costs among its various divisions. As part of its benchmarking efforts, AT&T compared the activity cost per driver unit for similar...
-
Access the May 1, 2002, filing of the fiscal 2002 10-K report for Dell Computer (ticker DELL) at www.SEC.gov. Read the footnote that details Dells segment information and answer the following: 1....
-
Homeowners Jim and Lisa Criss hired Kevin and Cathie Pappas, doing business as Outside Creations, to undertake a landscaping project. Kevin signed the parties contract as Outside Creations Rep. The...
-
A group of data items and their mean are given.. 15, 25, 35, 60, 90, 135 ; Mean = 60 a. Find the deviation from the mean for each of the data items. b. Find the sum of the deviations in part (a). a....
-
The following information is available for ADT Company, which produces special-order security products and uses a job order costing system. Overhead is applied using a predetermined overhead rate of...
-
Another Diphenhydramine synthesis is shown in the following equation: (a) Show a mechanism for the first step in this synthesis. (b) Explain which mechanism is occurring in the secondstep. OCH CH...
-
(a) Explain which of the following two products is formed when the reactant alkyl chloride reacts with sodium acetate in DMSO. (b) Explain which of the following two product alcohols is formed when...
-
What is the corridor principle? How does this corridor principle explain why the majority of business ideas are conceived at work?
-
Your firm anticipates receiving a $10 million payment in 3 months, and wants to invest that money for 3 months at ICELIBOR rates for 3 months (or 91 days) once they receive it. You contact your bank...
-
Watta Company has only common equity and bonds. You are asked to estimate Watta Company's WACC and you have the following information: Watta's next stock dividend is expected to be $1 per share. This...
-
Canadian tire located in waterloo is supposed to receive shipment of same product from Hamilton and Toronto. Shipment from Hamilton cost $0.2 per unit and shipment from Toronto costs $0.25 per unit....
-
c) Consider the following data: Amount ($) Common stock ($1 par value) 400,000 Capital surplus 900,000 Retained earnings 5,000,000 TOTAL OWNERS' EQUITY 6,300,000 Current market price of shares = $51...
-
The table below shows three European put options on the same underlying. Time To Expiration Exercise Price Option 1 3 months $65 Option 2 6 months $60 Option 3 6 months $65 Which put option is most...
-
Two auditors follow different approaches in determining sample size for unstratified difference estimation. The first auditor always selects a sample size of 75 items. He then calculates the...
-
Why can wastewater treatment requirements in Hawaii be less stringent than those in most locations on the U.S. mainland?
-
Solve each equation. 8 2 =x + 3 5 + 23 11 -x+ 5 45 6x + 7 3
-
Pentalene is a most elusive molecule and has never been isolated. The pentalene dianion, however, is well known and quite stable. Explain. 12- Pentalene Pentalene dianion
-
Indole is an aromatic heterocycle that has a benzene ring fused to a pyrrole ring. Draw an orbital picture of indole. (a) How many ? electrons does indole have? (b) What is the electronic...
-
Ribavirin, an antiviral agent used against hepatitis C and viral pneumonia, contains a 1, 2, 4-triazole ring. Why is the ringaromatic? 1,2,4-Triazole ring N- NH2 N-N Ribavirin OH
-
A: December 31, 2021, Southern Corporation reported the stockholders' equity accounts shown here (with dollar amounts in milions, except per-share amounts) (Click the icon to view the data)...
-
Part B Short Answer Questions Generally, each Part B short answer question would take about 250 words to answer adequately. Topic 4 (Foreign currency transactions) Sydney Ltd is an Australian company...
-
Concerning a RAL, which of the following is a true statement that needs to be discussed with the tax client? Question 16Select one: a. That the RAL is an "advanced" income tax refund b. A disclosure...

Study smarter with the SolutionInn App


