The energy level diagram for the MOs of CH 2 = CH 2 is shown if Figure
Question:
The energy level diagram for the MOs of CH2 = CH2 is shown if Figure 3.24. Show a similar diagram for the lowest-energy excited state of this molecule.
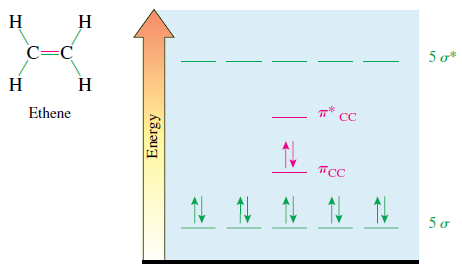
Transcribed Image Text:
н н C=C 5 o* н Н т* сс Ethene Псс 5 o Energy
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (18 reviews)
The lowest energy excited st...View the full answer

Answered By

ZIPPORAH KISIO LUNGI
I have worked on several other sites for more than five years, and I always handle clients work with due diligence and professionalism. Am versed with adequate experience in the fields mentioned above in which have delivered quality papers in research, thesis, essays, blog articles, and so forth.
I have gained extensive experience in assisting students to acquire top grades in biological, business and IT papers. Notwithstanding that, I have 7+ years of experience in corporate world software design and development.
5.00+
194+ Reviews
341+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show energy level diagram for the MOs of these compounds? a) H-C=N: b) H 0: C CH3 ) CHNH,
-
Show an energy level diagram for the MOs for He 2 and show how the electrons would be arranged in these MOs.
-
Figure 29.7 is an energy level diagram for 208Tl. What are the energies of the photons emitted for the six transitions shown? Figure 29.7 492 keV 472 keV 327 keV 40 keV
-
All adiabatic temperature changes occur as a result of a. changes in the absolute humidity of the air. b. expansion or compression of the air. c. changes is specific humidity d. the addition or...
-
The only variable overhead cost at Jackson Sporting Club is electricity. Does an unfavorable variable overhead spending variance imply that the club paid more than the anticipation per kilowatt hour?
-
Entry-level auditing staff often inspects client records and documentation supporting accounting transactions in order to gain evidence about the appropriate application of the applicable financial...
-
Prove the relations (11.16), (11.18), and (11.21).
-
The major classifications of activities reported in the statement of cash flows are operating, investing, and financing. Classify each of the transactions listed below as: 1. Operating activityadd to...
-
if a manager is paid a percentage of profits, does this generate a motive to manipulate accounts? would this be anticipated by principals and if so how would principals react to this expectation?
-
A new drug manufacturer wants to market a new drug only if he could be quite sure that the mean temperature of a healthy person taking the drug could not rise above 986 F, otherwise he will withhold...
-
Show energy level diagrams for the MOS of these compounds: a) H-C=C-CH3 b) CH0H c) CH3-C-H
-
Consider the species formed by the addition of an extra electron to H2 so that there are three electrons and a negative charge. Show an energy level diagram for the MOs of this species. Is there...
-
If credit cards were made illegal by government legislation, what would happen to velocity? Explain your answer.
-
Stillicum Corporation makes ultra light-weight backpacking tents. Data concerning the company's two product lines appear below: Direct materials per unit Direct labor per unit Direct labor-hours per...
-
Review given information below and create your own financial projections for the next 2 years for the honest company? COMPANY OVERVIEW The Honest Company is a personal care company in the United...
-
How do you make the plan happen? Who is responsible for each aspect of implementing the plan? b. What is the timing of your plan? Include time frames of when you should be most active based on the...
-
Discuss the environmental factors that have affected Dunkin', especially the competitive environment and the social and cultural environment, and how Dunkin' responded to a changing environment.
-
The marketing department has come up with the estimate that Applied Nanotech can sell 14 units per year at $306,000 net cash flow per unit for the next five years. The engineering department has come...
-
Explain the automatic disciplinary provisions of the AICPA and the actions by members that result in application of the provisions.
-
Wholesalers Ltd. deals in the sale of foodstuffs to retailers. Owing to economic depression, the firm intends to relax its credit policy to boost productivity and sales. The firms current credit...
-
Find the Maclaurin series of the function f(x) = e x and its radius of convergence.
-
How many H H eclipsing interactions would be present if cyclopentane were planar? Assuming an energy cost of 4.0kJ/mol for each eclipsing interaction, how much torsional strain would planar...
-
Two conformations of cis-1, 3-dimethylcyclobutanc is shown. What is the difference between them, and which do you think is likely to be more stable? (b) (a)
-
Draw two different chair conformations of cyclohexanol (hydroxycyclohexane), showing all hydrogen atoms. Identify each position as axial or equatorial.
-
Mahogany Company manufactures computer keyboards. The total cost of producing 15,000 keyboards is $430,000. The total fixed cost amounts to $130,000. Determine the total cost of manufacturing 25,000...
-
Assets Liabilities Common Stock Shareholders' Equity Retained Earnings $ In May, Rec Rooms, Inc., collected $8,000 owed by customers for services performed in April. Show the effect of this...
-
is federal legislation providing rules for how private-sector organizations collect, use, or disclose information about employees in the course of for-profit commercial activities in Canada. A)...

Study smarter with the SolutionInn App


