Upon consideration of the SiO2???Al2O3 phase diagram, Figure, for each pair of the following list of compositions,
Question:
Upon consideration of the SiO2???Al2O3 phase diagram, Figure, for each pair of the following list of compositions, which would you judge to be the more desirable refractory? Justify your choices.
(a) 20 wt% Al2O3-80 wt% SiO2 and 25 wt% Al2O3-75 wt% SiO2
(b) 70 wt% Al2O3-30 wt% SiO2 and 80 wt% Al2O3-20 wt%SiO2
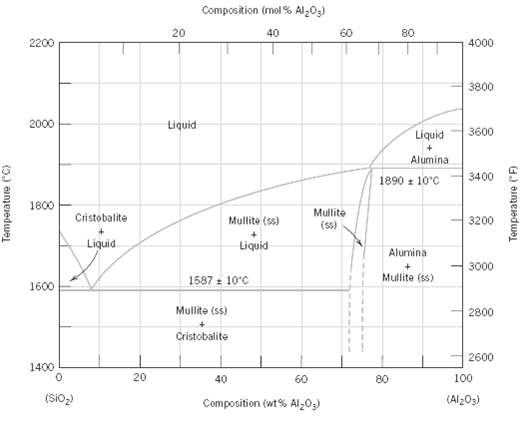
Transcribed Image Text:
Composition (mol % Al203) 80 20 40 60 2200 4000 3800 2000 Liquid Liquid 3600 Alumina 3400 1890 + 10°C 1800 Mullite (s) Cristobalite 3200 Mullite (ss) Liquid Liquid Alumina 3000 Mullite (ss) 1587 : 10°C 1600 Mulite (ss) 2800 Cristobalite 2600 1400 20 100 40 60 80 (SIO,) (Al203) Composition (wt % Al;03) Temperature ("C) Temperature ("F)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
a The 25 wt Al 2 O 3 75 wt SiO 2 will be more desirable because ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
For each pair of the following molecules, indicate whether its members are identical, structural isomers, conformers, or stereoisomers. How would you describe the relation between conformations when...
-
Upon consideration of the SiO2-Al2O3 phase diagram, Figure 12.27, for each pair of the following list of compositions, which would you judge to be the more desirable refractory? Justify your choices....
-
For each pair of the following compounds identify which compound would react more rapidly in an E1 reaction. a. b. CI .CI CI CI
-
Marsden Corp has developed a new strategic plan after rushing defective products to market hurt the company's sales and image. The strategic plan's initiatives are to focus on quality and develop...
-
Briefly describe the Religion of the Iceland. Briefly describe the Government of the Iceland. Briefly describe the Technology of the Iceland. How do these cultural elements described in this peer...
-
The Evolution of the Fashion Industry: Consider the market for clothes and suppose there exist 100 different styles that can be produced and can be arranged (and equally spaced) on a circle. Among...
-
Apex Electrical Supply, Inc., purchased inventory for \($2,000\) and also paid \($125\) freight to have the inventory delivered. Apex Electrical Supply, Inc., returned \($500\) of the goods to the...
-
Calculating EAR with Points you are looking at a one-year loan of $10,000. The interest rate is quoted as 9 percent plus three points. A point on a loan is simply 1 percent (one percentage point) of...
-
(a) Draw a payoff diagram of the company's equity and debt as a function of the company's asset value in 1 year (i.e. the maturity of the debt). (Draw them on the same graph) (b) What is the value of...
-
Sunn Co. manufactures a single product that sells for $180 per unit and whose variable costs are $135 per unit. The companys annual fixed costs are $562,500. 1. Prepare a contribution margin income...
-
Find the maximum temperature to which the following two magnesia???alumina refractory materials may be heated before a liquid phase will appear. (a) A spinel-bonded alumina material of composition 95...
-
Compute the mass fractions of liquid in the following refractory materials at 1600C (2910F): (a) 6 wt% Al2O3-94 wt% SiO2 (b) 10 wt% Al2O3-90 wt% SiO2 (c) 30 wt% Al2O3-70 wt% SiO2 (d) 80 wt% Al2O3-20...
-
Comparative data from the balance sheet of Dressaire Inc. are shown below. Instructions (a) Perform a horizontal percent of base year analysis using 2015 as the base year. (b) Calculate the...
-
1. How does business development change as the PLC progresses through its stages? 2. List the roles and responsibilities of the three different bands in the management of simultaneous life cycles. 3....
-
Jobs At Whole Foods (1) Job A Kind of Work Provide excellent customer service. Follow and comply with all applicable health and sanitation procedures. Prepare food items: make sandwiches; slice deli...
-
large hollow cylinder of mass M stays on a horizontal plane. A small body of mass m starts to move from the point B on the surface of the cylinder towards the highest point A in such a way that it is...
-
How does HR Planning differ for different types of organizations, such as small businesses, multinational corporations, and non-profits?
-
How does the integration of automation and Industry 4.0 technologies impact the efficiency and productivity of manufacturing processes?
-
Two protons are fired toward each other on closely spaced paths, one moving in the \(+z\) direction and one in the \(-z\) direction. As they pass close to each other, is the magnetic force between...
-
Suppose you are comparing just two means. Among the possible statistics you could use is the difference in means, the MAD, or the max min (the difference between the largest mean and the smallest...
-
Sketch a graph of the potential energy of two atoms as a function of the distance between them. On your graph, indicate how bond energy and bond distance are defined.
-
What is the difference between a coherent precipitate and a distinct second-phase particle?
-
What is overaging?
-
What types of heating and cooling conditions are imposed in an I-T or T-T-T diagram? Are they realistic for the processing of commercial items?
-
An initially motionless test car is accelerated uniformly to 120 km/h in 7.98 s before striking a simulated deer. The car is in contact with the faux fawn for 0.515 s, after which the car is measured...
-
How do you define operational excellence? What factors are involved in achieving operational excellence? Who (within an organization) is responsible for operational excellence and why is this...
-
f(x) = x In(2x) Find f(x) for the function below....

Study smarter with the SolutionInn App


