Vapor-liquid equilibrium data at 101.3 kPa are given for the chloroform-methanol system. From these data, prepare plots
Question:
Vapor-liquid equilibrium data at 101.3 kPa are given for the chloroform-methanol system. From these data, prepare plots like Figures 4.6b and 4.6c. From the plots, determine the azeotropic composition and temperature at 101.3 kPa. Is the azeotrope of the minimum- or maximum-boilingtype?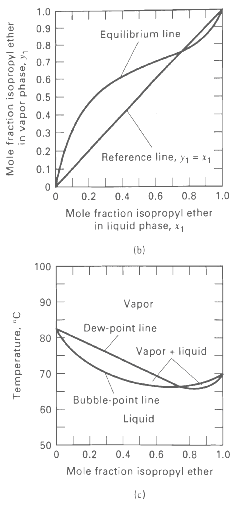
Transcribed Image Text:
1.0 0.9 Equilibrium line 0.8 0.7E 0.6E 0.5 0.4 0.3 0.2 Reference line, y1 = 11 0.1 F 0.6 0.8 1.0 0.2 0.4 Mole fraction isopropyl ether in liquid phase, x1 (b) 100 90 Vapor Dew-point line 80 Vapor • liquid 70 Bubble-point line 50 Liquid 50 0.2 0.4 0.6 0.8 1.0 Mole fraction isopropyl ether Mole fraction isopropyl ether in vapor phase, y, Temperature, "C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
See plots below From these plots a minimumboiling azeo...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
The following equations are given for the liquid-phase activity coefficients of the water (W)-acetic acid (A) system. Find the dew point and bubble point of a mixture of composition xw = 0.5, xA =...
-
Frequency tables are given for the first 100 digits in the decimal representation of 77 and the first 100 digits in the decimal representation of 22/7. a. Construct histograms representing the...
-
Liquid water at 200 kPa and 15C is heated in a chamber by mixing it with superheated steam at 200 kPa and 150C. Liquid water enters the mixing chamber at a rate of 4.3 kg/s, and the chamber is...
-
Google, the immensely popular Web search engine, has been touted as the closest thing the Web has to an ultimate answer machine. Although this is debatable, of course, it is far more difficult to...
-
An aging analysis of Reiko Limited's accounts receivable at December 31, 2012 and 2011 showed the following: 1. At December 31, 2011, the unadjusted balance in Allowance for Doubtful Accounts was a...
-
Use the data visualization methods presented in this chapter to explore these data and discover relationships between the variables. Include the following, in your report: 1. Create a scatter chart...
-
Investment Motives of Firms. What is the difference between proactive and defensive investment motives?
-
Telfer, Inc. reported net income of $2.8 million in 2017. Depreciation for the year was $160,000, accounts receivable decreased $350,000, and accounts payable decreased $280,000. Compute net cash...
-
1. A wire of length 50 units is cut into two pieces, somewhere along the wire. One piece is shaped into a square, and the other is shaped into a circle. What is the maximum possible value of the...
-
Richard Pitkin, CFO of Draper Corporation, was concerned by the long-term prospects for the Synectics product line. The product line had performed well historically, but the impending loss of its...
-
Using vapor pressure data from Exercises 4.6 and 4.8 and the enthalpy data provided below: (a) Construct an h-x-y diagram for the benzene-toluene system at 1 atm (101.3 kPa) based on the use of...
-
Vapor-liquid equilibrium data at 101.3 kPa are given for the water-formic acid system. From these data, prepare plots like Figures 4.7b and 4.7c. From the plots, determine the azeotropic composition...
-
Use a change of variables to evaluate the following definite integrals. r2/5 dx 2/(5V3) xV25x2 1
-
Explain how crossover, mutation, and reproduction affect schemata.
-
Describe how to strategically select, prioritize, and resource projects.
-
Explain how schemata help us to understand why genetic algorithms work. What does the schema theorem tell us?
-
Explain why introducing predators can help in systems that use artificial evolutionary techniques.
-
Develop project scope based on conceptualization of the project.
-
Which of the following situations would not support the auditor's decision to reduce control risk below maximum for the audit of intangible assets? a. The client employs a qualified specialist who...
-
Explain how the graph of each function can be obtained from the graph of y = 1/x or y = 1/x 2 . Then graph f and give the (a) Domain (b) Range. Determine the largest open intervals of the domain over...
-
Discuss various investment roles played by ETFs.
-
A vaporliquid mixture at 250F and 500 psia contains N 2 , H 2 S, CO 2 , and all the normal paraffins from methane to heptane. Use Figure 2.4 to estimate the K-value of each component. Which...
-
One thousand kmol/h of rich gas at 70F with 25% C 1 , 15% C 2 , 25% C 3 , 20% nC 4 , and 15% nC 5 by moles is to be absorbed by 500 kmol/h of nC 10 at 90F in an absorber at 4 atm. Calculate by the...
-
When calculating multicomponent distillation, why is it best to list the components in order of decreasing volatility? In such a list, do the two key components have to be adjacent?
-
1 ABC Corp 2 Trial Balance 3 December 31, 2022 and 2021 4 5 B 2 D E Year 2021 Cr Db Cr Year 2022 Db Prot F G H To c this 69 7 Cash $14,800 $ 6,375 8 Accounts Receivable 13,225 91,625 9 Building...
-
Barnett Brass sells economy door knobs for $15 each. Unit product costs are as follows: Direct materials Direct labor Manufacturing overhead Total $ 3 2 6 $11 An order to purchase 4,000 units was...
-
Culver Inc. presented the following data. Net income $2,610,000 Preferred stock: 51,000 shares outstanding, $100 par, 7% cumulative, not convertible 5,100,000 Common stock: Shares outstanding 1/1...

Study smarter with the SolutionInn App


