We?ll see that organic molecules can be classified according to the functional groups they contain, where a
Question:
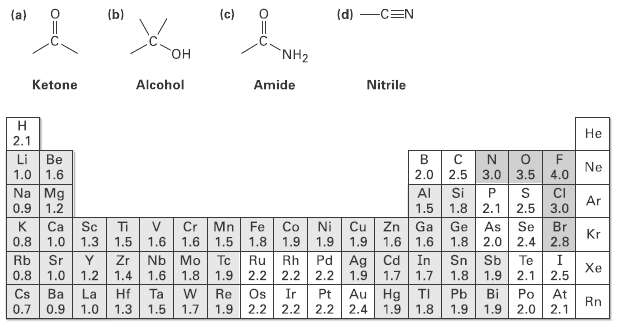
We?ll see that organic molecules can be classified according to the functional groups they contain, where a functional group is a collection of atoms with a characteristics chemical reactivity. Use the electro negativity values given in Figure to predict the direction of polarization of the following functional groups.
Transcribed Image Text:
(a) (b) (c) (d) -CEN NH2 HO Ketone Alcohol Amide Nitrile н Не 2.1 Li Be в Ne 3.0 3.5 Si 1.0 1.6 2.0 2.5 4.0 Na Mg 1.2 AI CI Ar 1.8 2.1 3.0 0.9 1.5 2.5 Sc Ti V Cr Mn Fe Co 1.6 1.3 Zn Ga 1.6 Ca Ni Cu Ge Se Br As 2.0 Kr 0.8 1.0 1.5 1.6 1.5 1.8 1.9 1.9 1.9 1.6 1.8 2.4 2.8 Pd Ag | Cd In Rb 0.8 Sr Zr Nb Mo 1.8 Tc Ru Rh 2.2 2.2 Sn Sb 1.9 Te Xe 1.0 1.2 1.4 1.6 1.9 2.2 1.9 1.7 1.7 1.8 2.1 2.5 Au Hg Cs La Re Os Ir 2.2 2.2 2.2 Pb Ba 0.9 Hf 1.3 Ta 1.5 Pt TI 1.9 1.8 Bi 1.9 Po 2.0 At 2.1 Rn 0.7 1.0 1.7 1.9 2.4 1.9
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
00 C b ...View the full answer

Answered By

Bharat bhushan Kumar
I have been providing coaching for Students at India for various competitive exams such as IIT-JEE AND NEET! I excell at mathematics as i won NMTC IN MY HIGH SCHOOL! My speciality is in Algebra, calculus, Number theory, BASIC PHYSICS
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain how products can be classified according to the spending behaviors and give examples .
-
Reactions that involve PLP can be classified according to which bond to the alpha carbon is broken. For the amino acid-PLP adduct shown below, indicate what kind of reaction involves cleavage of...
-
Inherent risks can be classified according to their nature, such as unusual pressure on management or account likely to require adjustments; and also as either a financial report level inherent risk...
-
There are three general methods of allocating overhead costs: plantwide rates, rates for each expense category, and departmental rates. Describe when each is the most useful.
-
In 2016, Henri, a U.S. citizen and calendar year taxpayer, reports $30,000 of income from France, which imposes a 10% income tax, and $50,000 from Italy, which imposes a 40% tax. In addition, Henri...
-
Briefly describe the composition and general thrust of each of the following indexes. a.NYSE composite index b.Amex composite index c.Nasdaq Stock Market indexes d.Value Line composite index
-
Amy Company is considering two alternatives. Alternative A will have sales ol \($160,000\) and costs of \($100,000\). Alternative B will have sales of \($180,000\) and costs ol \($125,000\). Compare...
-
Brazilia Corporation needed a warehouse and maintenance facility on its company site, which already housed three manufacturing/ storage facilities and the company head office. The lowest outside bid...
-
DEPRECIATION FACTS DEPRECIATION TEMPLATE DEPRECIATION-JE TEMPLATE STUDENT NAME ACCOUNTING 2113 (FINANCIAL ACCOUNTING) DEPRECIATION CALCULATIONS CHERYLL'S FURNITURE COMPANY Asset Acquisition For the...
-
Plant, Inc., is considering making an offer to purchase Palmer Corp. Plant's vice president of finance has collected the following information: Plant also knows that securities analysts expect the...
-
Carbocations, ions that contain a trivalent, positively charged carbon atom, react with water to give alcohols. How can you account for the fact that the following carbocation gives a mixture of two...
-
Phenol, C6H5OH, is a stronger acid then methanol, CH3OH, even though both contains an O ? H bond. Draw the structures of the anions resulting from loss of H+ from phenol and methanol, and use...
-
Hot Lunch Delivery Service has always had a policy to pay stockholders annual dividends in an amount exactly equal to net income for the year. Joe Alberg, the companys president, is confused because...
-
An entity sells goods under warranty and past experience shows that minor defects account for 10% of sales and major defects account for 2% of sales. If minor defects were detected in all goods sold...
-
Manufacturing profits" is referred to as "creative accounting" or earnings management. How can the directors' go about "manufacturing profits" without actually improving the real profitability of the...
-
The newspaper reported last week that Jernigan Enterprises earned $34.14 million this year. The report also stated that the firm's return on equity is 13 percent. The firm retains 70 percent of its...
-
A galaxy known to be traveling away from the Earth at 0 . 7 0 0 percent of the speed of light is observed on Earth to have a strong peak in its spectrum at 6 3 3 . 1 nm . However, one small portion...
-
In what ways do organizational structures and systems adapt and evolve to accommodate the dynamic demands of change initiatives ?
-
Following are the net earnings of four large companies for the fiscal years from 1999 to 2002. These amounts are in thousands of dollars. Required: Based on this information alone, decide which of...
-
In the circuit shown in Figure 4, a battery supplies a constant voltage of 40 V, the inductance is 2 H, the resistance is 10, and l(0) = 0. (a) Find l(t). (b) Find the current after 0.1s.
-
The Joule-Thomson coefficient, , given by is a function of temperature. The temperature at which = 0 is known as the inversion temperature. a. Use the van der Waals equation of state to determine...
-
(a) Show all of the steps in the mechanism for this reaction. Don't forget to use curved arrows to show the movement of electrons in each step of the mechanism. (b) Show a free energy versus reaction...
-
Which of these compounds would have a faster rate of SN2 reaction? a) b) c) d) e) CI CI Br or or Br or Br or or CI CI Br Br
-
Which of these compounds would have faster rate of SN1 reaction? a) b) c) d) e) CI ta Ph Ph -CI or Br or Br or or -Cl or + CH3 Ph -Cl Br Br ta -CI
-
Exercise 10-19B Effective Interest: Amortization of bond premium LO P6 Quatro Co. issues bonds dated January 1, 2019, with a par value of $860,000. The bonds' annual contract rate is 10%, and...
-
QUESTION 1 Machinery was purchased 3 years ago for $542,000. Depreciation is calculated 15% and 25% straight-line for accounting purposes and tax purposes respectively. Calculate the Carrying Amount...
-
Required: 1. Determine each year's absorption costing operating income. Present your answer in the form of a reconciliation report. Absorption Costing Operating Incomes Year 1 Year 2 Year 3 Variable...

Study smarter with the SolutionInn App


