What carbonyl compounds would you reduce to prepare the following alcohols? List all possibilities. (b)
Question:
What carbonyl compounds would you reduce to prepare the following alcohols? List all possibilities.
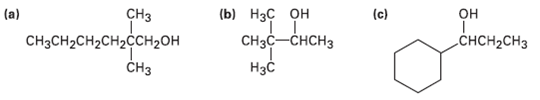
Transcribed Image Text:
(b) Нзс он Cнзс—снсHз Нас |(a) (c) СHз CHзCH2CH2сH2ссH2он CНз он CHCH-CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (13 reviews)
a b c Alcohol CH3 CH3CHCHCHCCHOH ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What alkenes might be used to prepare the following alcohols by hydroboration/oxidation? (a) CH3 (b) CHH (c) .CH- CHCHCH2CH2OH
-
What reagents would you use to prepare the following compounds? a. b. CH CCH CH:C 0 CH3CCH-CH-CH (COCH-CH3 )2
-
What alkyl halides would you use to prepare the following ketones by an acetoacetic ester synthesis? CH (a) (b) CHH,H,H3 -CH-CH2CH2CCH
-
Chambers Corporation purchased a piece of equipment for $36,000. It estimated a 6-year life and $6,000 salvage value. Thus, straight-line depreciation was $5,000 per year [($36,000 $6,000) 6]. At...
-
Describe SWOT analysis as a way to guide internal analysis. How does this approach reflect the basic strategic management process?
-
Briefly describe each of the following disbursement products/methods: Zero-balance accounts (ZBAs) Controlled disbursement Positive pay How does a ZBA relate to the firms target cash balance?
-
What would be a forensic accounting certificate from the AICPA? a. Cr. FA b. CFF c. CFFA d. CFS e. CVA
-
Martin Shoes, Inc. manufactures and distributes orthopedic footwear. To sell its products, the marketing department requires sales personnel to call on the shoe retailers within their assigned...
-
DISCUSSION ACTIVITY The Canadian Human Rights Commission oversees both the Canadian Human Rights Act and the Employment Equity Act. Briefly describe in your own words the purpose of each o...
-
Hatcher Enterprises uses a chemical called Rbase in production operations at five divisions. Only six suppliers of Rbase meet Hatchers quality control standards. All six suppliers can produce Rbase...
-
What Grignard reagent and what carbonyl compound might you start with to prepare the following alcohols? CH (a) (b) (c) CHCH-CHCH2CH CHH2H H2c=C "CH- (d) (e) (f) .CH- "
-
How would you carry out the following transformations? Co .CO2H (a) (b) Co CH2 C (c) CH2SH
-
If you wish to cook a piece of meat thoroughly on an open fire, why should you not use a high flame? (Note that carbon is a good thermal insulator.)
-
General Information and Confidential Instructions for the Environmental League Negotiator (from the League's Board of Directors) A newly formed national consortium, Harborco, is interested in...
-
1. Discuss the strategies of a couple of U.S. iconic oligopolists, and reflect on their distinctive features and strategies. 2. Examine the whole range of pricing strategies that an oligopolist can...
-
In a certain antibacterial manufacturing process, the average percentage of alcohol in the gel is 79.012% and its standard deviation is 0.545%. It has been determined that the nominal value of the...
-
How does applied behavior analysis relate to forensic psychology? How does applied behavior analysis not relate to forensic psychology?
-
It is very important to begin an investigation promptly once an employer has received a complaint, particularly in instances involving sexual harassment. Saxton, Bennett , and Steiner, are three...
-
Ensign Corporation is a manufacturing firm with ten domestic plants. Increased demand for the company's products and a near full capacity production have caused management to decide to build a new...
-
In the figure, two loudspeakers, separated by a distance of d1 = 2.63 m, are in phase. Assume the amplitudes of the sound from the speakers are approximately the same at the position of a listener,...
-
In a capillary rise experiment, the height (h) to which a liquid rises depends on the density (d) and surface tension () of the liquid and the radius of the capillary (r). The equation relating these...
-
Chloramphenicol (at right) is a potent antibiotic, isolated from Streptomyces venezuelae, that is particularly effective against typhoid fever. It was the first naturally occurring substance shown to...
-
(a) Give the (R,S) designations for each chirality center in compound A and for compound B. (b) Write the Fischer projection formula for a compound C that is the diastereomer of A and B. (c) Would C...
-
(a) Is trans-1, 2-dimethylcyclopentane (5) superposable on its mirror image (i.e., on compound 6)? (b) Is cis-1, 2-dimethylcyclopentane (7) superposable on its mirror image? (c) Is cis-1,...
-
What is one purpose of the symbolism used in this passage? A. To illustrate the destruction of Granny's dream B. To make a connection between Granny and the reader C. To link two unlike concepts: a...
-
Payroll taxes levied against employees become liabilities a. at the end of an accounting period Ob. when data are entered in a payroll register c. on the first of the following month Od. at the end...
-
Regarding inventory, center directors should Group of answer choices keep a list of items that the center director could not replace keep a record of at least the major items purchased keep a record...

Study smarter with the SolutionInn App


