According to the Einstein model of a crystalline solid, the internal energy per mole is given by
Question:
According to the Einstein model of a crystalline solid, the internal energy per mole is given by
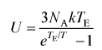
where TE is a characteristic temperature called the Einstein temperature, and T is the temperature of the solid in kelvins. Evaluate the molar internal energy of diamond (TE = 1060 K) at 300 K and 600 K, and thereby the increase in internal energy as diamond is heated from 300 K to 600 K.
Transcribed Image Text:
3N KT e -1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (18 reviews)
We determine U 300 and U ...View the full answer

Answered By

Felix Mucee
I am a detailed and thorough professional writer with 5 years of administrative experience- the last 2 years in academic writing and virtual office environment. I specialize in delivering quality services with respect to strict deadlines and high expectations. I am equipped with a dedicated home office complete with a computer, copier/scanner/fax and color printer.
I provide creative and detailed administrative, web search, academic writing, data entry, Personal assistant, Content writing, Translation, Academic writing, editing and proofreading services. I excel at working under tight deadlines with strict expectations. I possess the self-discipline and time management skills necessary to have served as an academic writer for the past five years. I can bring value to your business and help solve your administrative assistant issues.
4.70+
13+ Reviews
33+ Question Solved
Related Book For 

Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Question Posted:
Students also viewed these Thermodynamics questions
-
Describe the distinguishing characteristics of a crystalline solid and an amorphous solid.
-
Think of a crystalline solid as a set of atoms connected by ideal springs. When a wire is stretched, how is the elongation of the wire related to the elongation of each of the interatomic springs?...
-
A gas at 350 K and 12 atm has a molar volume 12 per cent larger than that calculated from the perfect gas law. Calculate (a) The compression factor under these conditions and (b) The molar volume of...
-
(a) Show that (x) = 2x + 3x 36x is not one-to-one on (-, ). (b) Determine the greatest value c such that is one-to-one on (-c, c).
-
Stahl Company is conducting a time-driven activity-based costing study in its Shipping Department. To aid the study, the company provided the following data regarding its Shipping Department and the...
-
Friedman, the "O" Street Carpet Shop, Inc., and Langness formed a partnership known as NFL Associates. "O" Street Carpet's net contribution to capital was $5,004; Langness contributed $14,000 in...
-
During its first year of operation, Krabna Company purchased 5,600 units of a product at $42 per unit. During the second year, it purchased 6,000 units of the same product at $48 per unit. During the...
-
Quester Company operates a large discount store and uses the retail method to estimate the cost of ending inventory . Management suspects that in recent weeks there have been unusually heavy losses...
-
The hypotenuse of a right triangle has one end at the origin and one end on the curve y(x) = xe-4X, with x 2 0 . One of the other two sides is on the x-axis, the other side is parallel to the y-axis....
-
Ellipses Corp is a small business that operates in Herndon, VA. The company is located at10 Period Lane, Herndon, VA 20170. Its federal Employer Identification Number is 77-7777777, and its...
-
A vertical heat-insulated cylinder is divided into two parts by a movable piston of mass m. Initially the piston is held at rest. The top part is evacuated and the bottom part is filled with 1 mole...
-
In an isothermal expansion, an ideal gas at an initial pressure P0 expands until its volume is twice its initial volume. (a) Find its pressure after the expansion. (b) The gas is then compressed...
-
A job shop commenced its operations on January 1 of the current year. During the first quarter, the following transactions took place: (i) Materials costing 4,00,000 were purchased on account. (ii)...
-
Cash receipt from sale of equipment $ 17,000 Depreciation expense 13,000 Cash payment of dividends 5,800 Cash receipt from issuance of common stock 20,000 Net income 38,000 Cash purchase of land...
-
1a. List 5 benefits and five (5) challenges of performance assessment? 1b. Link each benefit to the impact on the organization AND the individual? 1c. In module one you identified the link between...
-
CoursHeroTranscribedText Quilcene Oysteria farms and sells oysters in the Pacific Northwest. The company harvested and sold 7,500 pounds of oysters in August. The company's flexible budget for August...
-
On July 1, Davidson Corporation had the following capital structure: Common stock ($4 par value) Additional paid-in capital Retained earnings Treasury stock Required: $ 678,000 960,000 840,000...
-
A partially consumed keg of beer has 6 gallons and 36 fluid ounces remaining. Express this volume in terms of a. fluid ounces b. gallons (the answer should be a mixed number with the fraction in the...
-
The Arizona Corporation reported positive net income but negative cash flow from operations. Further, despite the negative cash flow from operations, the company continued to pay its regular dividend...
-
Consider the setup in Problem 16. Show that the relative speed of the ball and the point of contact on the stick is the same before and immediately after the collision. (This result is analogous to...
-
Calculate the molar solubility of Mg(OH) 2 in 1.00 M NH 4 Cl(aq).
-
Two small spheres with mass m = 15.0 g are hung by silk threads of length L = 1.20 m from a common point (Fig. 21.44). When the spheres are given equal quantities of negative charge, so that q1 = q2...
-
Two identical spheres are each attached to silk threads of length L = 0.500 m and hung from a common point (Fig. 21.44). Each sphere has mass m = 8.00 g. The radius of each sphere is very small...
-
Sodium chloride (NaCl, ordinary table salt) is made up of positive sodium ions (Na+) and negative chloride ions (c1-). (a) If a point charge with the same charge and mass as all the Na + ions in...
-
The following information relates to labor for Bridgeport Appliance Repair Shop. \ table [ [ Repair - technicians' wages,$ 1 4 0 , 0 0 0
-
Current Attempt in Progress The following three accounts appear in the general ledger of Bonita Corp. during 2 0 2 2 . \ table [ [ , Equipment,,, ] , [ Date , Balance,Debit,Credit,Balance ] , [ Jan ....
-
Xonic Corporation issued $ 8 million of 2 0 - year, 8 percent bonds on April 1 , 2 0 2 1 , at 1 0 2 . Interest is paid on March 3 1 and September 3 0 of each year, and all of the bonds in the issue...

Study smarter with the SolutionInn App


