Consider the arrangement of the three black surfaces shown, where A 1 is small compared to A
Question:
Consider the arrangement of the three black surfaces shown, where A1 is small compared to A2 or A3. Determine the value of F13. Calculate the net radiation heat transfer from A1 to A3 if A1 = 0.05 m2, T, = 1000 K, and T3 = 500 K.
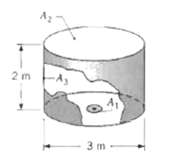
Transcribed Image Text:
3 m
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (19 reviews)
KNOWN Arrangement of three black surfaces with prescribed geometries and sur...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Heat and Mass Transfer
ISBN: 978-0471457282
6th Edition
Authors: Incropera, Dewitt, Bergman, Lavine
Question Posted:
Students also viewed these Mechanical Engineering questions
-
Consider the arrangement of charges described in Exercise 23.23. (a) Derive an expression for the potential V at points on the y-axis as a function of the coordinate y. Take V to be zero at an...
-
Consider the arrangement of point charges described in Exercise 23.25. (a) Derive an expression for the potential V at points on the y-axis as a function of the coordinate y. Take V to be zero at an...
-
Consider the arrangement of bulbs shown in the drawing. Each of the bulbs contains a gas at the pressure shown. What is the pressure of the system when all the stopcocks are opened, assuming that the...
-
The Kelly Services, Inc., and Subsidiaries statements of earnings from its 2008 annual report are presented in Exhibit 5-4. Required a. Using the statements of earnings, prepare a vertical...
-
Explain the differences between high- and low-context cultures, giving some examples. What are the differential effects on the communication process?
-
A sphere completely submerged in water is tethered to the bottom with a string. The tension in the string is one-third the weight of the sphere. What is the density of the sphere?
-
(d) Compare the estimate of the variance component for the term family with its SE. Does their relative magnitude suggest that this term might reasonably be dropped from the model?
-
As a result of the recent mortgage crisis, many banks reported record losses to their mortgage receivables and other assets based on the decline in these assets fair values. Requirements 1. What...
-
Current Attempt in Progress Thomas - Britt Industries was founded by Matt Thomas in 1 9 5 0 as a small machine shop that produced parts for the aircraft industry. The Korean War brought rapid growth...
-
Lucie likes consuming candy (c) and fruit (f), and dislikes consuming plastic packaging (p), and has rational preferences over bundles (c, f, p). No matter how many units of each good she has, Lucie...
-
A drying oven consists of a long semicircular duct of diameter D = 1 m. Materials to be dried cover the base of the oven while the wall is maintained at 1200 K. What is the drying rate per unit...
-
A long, V-shaped part is heat treated by suspending it in a tubular furnace with a diameter of 2 m and a wall temperature of 1000 K. The "vee" is I-m long on a side and has an angle of 60?. If the...
-
Describe the differences between mass production and custom production of goods and services. Explain how these differences influence the costing method.
-
Silver Company makes a product that is very popular as a Mothers Day gift. Thus, peak sales occur in May of each year, as shown in the companys sales budget for the second quarter given below: April...
-
Among the following statements, select the ones which have a positive environmental impact. Choose several answers Minimising the impact of a product on the environment Avoiding the destruction of a...
-
Developing Financial Statements: All organizations, including those in the healthcare industry, need to make money to be profitable and survive. Financial statements, such as balance sheets, profit...
-
The engineers estimated that on average, fuel costs, assuming existing routes and number of flights stay the same, would decrease by almost 18% from an average of 42,000 gallons of jet fuel per...
-
It's the latest Berkeley trend: raising chickens in a backyard co-op coop. (The chickens cluck with delight at that joke.) It turns out that Berkeley chickens have an unusual property: their weight...
-
The company position in the industry. LO.1
-
Identify the source of funds within Micro Credit? How does this differ from traditional sources of financing? What internal and external governance mechanisms are in place in Micro Credit?
-
If the internal energy of the products of a reaction is higher than the internal energy of the reactants, what is the sign of E for the reaction? In which direction does energy flow?
-
Flow through the converging nozzle in Fig. P4.2 can be approximated by the one-dimensional velocity distribution u ≈ Vo (1 + 2x/L) v ≈ 0 w ≈ 0 (a) Find a general expression for the...
-
A two-dimensional velocity field is given by in arbitrary units. V = (x2 y2 + x)i (2xy + y)j At (x, y) = (1, 2), compute (a) The accelerations ax and ay, (b) The velocity component in the direction...
-
Suppose that the temperature field T = 4x2 3y3, in arbitrary units, is associated with the velocity field of Prob. 4.3. Compute the rate of change dT/dt at (x, y) = (2, 1).
-
Caspian Sea Drinks needs to raise $74.00 million by issuing additional shares of stock. If the market estimates CSD will pay a dividend of $2.69 next year, which will grow at 3.45% forever and the...
-
i need help in B and C Integrative Case 5-72 (Algo) Cost Estimation, CVP Analysis, and Decision Making (LO 5-4.5.9) Luke Corporation produces a variety of products, each within their own division....
-
Relate PSA (Public Securities Association) speed to the average life of a MBS. Describe the PSA measure and discuss which MBS would have the greater average life, one with a PSA of 100 or one with a...

Study smarter with the SolutionInn App


