Question: Determine the diffusivity of H2S in water, using the penetration theory, from the following data for the absorption of H2S into a laminar jet of
Determine the diffusivity of H2S in water, using the penetration theory, from the following data for the absorption of H2S into a laminar jet of water at 20?C. Jet diameter = 1 cm, Jet length = 7 cm, and Solubility of H2S in water = 100 mol/m3 The average rate of absorption varies with the flow rate of the jet asfollows:
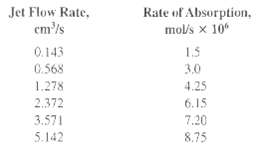
Jet Flow Rate, em'/s Rate of Absorption, mol's x 10 1.5 0.143 0.568 3.0 1.278 4.25 2.372 6.15 7.20 3.571 5.142 8.75
Step by Step Solution
3.42 Rating (177 Votes )
There are 3 Steps involved in it
Use cgs units From a rearrangement of Eq 3192 Surface a... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
37-E-C-E-S-P (86).docx
120 KBs Word File


