Figure shows a cycle consisting of five paths: AB is isothermal at 300 K, BC is adiabatic
Question:
Figure shows a cycle consisting of five paths: AB is isothermal at 300 K, BC is adiabatic with work = 5.0 J, CD is at a constant pressure of 5 atm, D E is isothermal, and EA is adiabatic with a change in internal energy of 8.0 J. What is the change in internal energy of the gas along pathCD?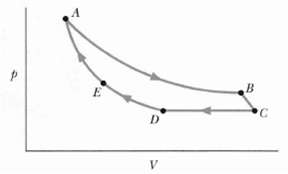
Transcribed Image Text:
D. V
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
For convenience the int subscript for the internal energy will be omitted in this solution R...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Question Posted:
Students also viewed these Thermodynamics questions
-
Figure shows a cycle undergone by 1.00 mol of an ideal monatomic gas. The temperatures are T1 = 300 K, T2 = 600 K, and T3 = 455 K. For 1 ? 2, what are?(a) Heat Q,(b) The change in internal energy...
-
An ideal gas goes through a cycle consisting of alternate isothermal and adiabatic curves (Fig. 2.2). The isothermal processes proceed at the temperatures T1, T2, and T3. Find the efficiency of such...
-
An ideal gas goes through a cycle consisting of isothermal, polytropic, and adiabatic lines, with the isothermal process proceeding at the maximum temperature of the whole cycle. Find the efficiency...
-
Explain the advantages of using JDBC metadata for Java database applications.
-
The Becker Company factory has been experiencing long delays in jobs going through the turret lathe department because of inadequate capacity. The head of this department contends that five machines...
-
The following state transition table is a simplified model of process management, with the labels representing transitions between states of READY, RUN, BLOCKED, and NONRESIDENT. Give an example of...
-
Visit the Center for Internet Security Web site (www.cisecurity.org) and down load the appropriate scoring tool for your computer. Run the tool to assess how well your computer conforms to the...
-
Sandy Dawson is the Chief Operating Officer at Mercy Hospital in Atlanta, Georgia. She is analyzing the hospital's overhead costs but is not sure whether nursing hours or the number of patient days...
-
Fedral Court Appel: Hilewitz v. Canada (Minister of Citizenship & Immigration) Judicial Reasoning 1. In explaining their position, the FCA reviews the legislative history and jurisprudence related to...
-
Helium gas with a volume of 2.60 L, under a pressure of 1.30 atm and at a temperature of 41.0oC, is warmed until both pressure and volume are doubled. (a) What is the final temperature? (b) How many...
-
(a) What is the number of molecules per cubic meter in air at 20oC and at a pressure of 1.0 atm (= 1.01 x 105 Pa)? (b) What is the mass of 1.0 m3 of this air? Assume that 75% of the molecules are...
-
An ideal gas initially at 300 K is compressed at a constant pressure of 25 N/m2 from a volume of 3.0 m3 to a volume of 1.8 m3. In the process, 75 J is lost by the gas as heat. What are? (a) The...
-
Supply compatible hashCode and equals methods to the Student class described in Exercise P15.2 . Test the hash code by adding Student objects to a hash set. Data from exercise P15.2 Reimplement...
-
what an enterprise agreement is the principles of enterprise agreements that must be applied in maintaining payroll systems. 2. Identify and briefly describe one (1) other industrial instruments...
-
1. From the HRM perspective, what are the implications of managing labor relations in the Public Sector? 2. Discuss whether unions are still relevant and necessary in today's work environment. What...
-
Ballard corporation's stock returns for years 1 - 4 were 20.00%, 15.00%, 10.00% and 5.00%.Given this information, calculate the standard deviation of the returns.
-
List key ways in which would make the most use of the cost analysis in supporting VectorCal's contract with the federal government.What would happen if the cost analysis related to the VectorCal's...
-
1. Do you feel differently about our criminal justice system after watching the movie? 2. Do you agree with Stevenson's lament that "wealth, not culpability, shapes outcomes" in our justice system?...
-
Under atmospheric conditions the acetone-chloroform azeotrope boils at \(64.6^{\circ} \mathrm{C}\) and contains 33.5 mole percent acetone. The vapour pressures of acetone and chloroform at this...
-
Describe a group you belong or have belonged discuss the stages of group development and suggest how to improve the group effectiveness by using the group development model.
-
Did cycles of boom and bust exist under feudalism? Why or why not?
-
The aircraft carrier John F. Kennedy has mass 7.4 X 107 kg. When its engines are developing their full power of 280,000 hp, the John F. Kennedy travels at its top speed of 35 knots (65 km/h). If 70%...
-
A ski tow operates on a 15.0" slope of length 300 m. The rope moves at 12.0 km/h and provides power for 50 riders at one time, with an average mass per rider of 70.0 kg. Estimate the power required...
-
A typical flying insect applies an average force equal to twice its weight during each downward stroke while hovering. Take the mass of the insect to be 10 g, and assume the wings move an average...
-
You applied for the following student load to support your college study: Before graduation: You will receive $8000 /per year from the beginning of each school year for 4 years; You will graduate...
-
A decision maker with an exponential u-function and a risk aversion coefficient, y= 0.001, who is engaging in a sealed bid auction. The u-curve is, U(x)=1-exp(-yx) The decision maker's PIBP for the...
-
Southwest Airlines Recent stock price $ 20.69 P/E ratio 49.3 Dividend yield 0.10% Beta 1.1 Financial statement forecast 2001 2002E 2003E 2005E/2007E Total debt ($ mill) 1841.8 Revenue ($ mill) 5555.2...

Study smarter with the SolutionInn App


