Figure shows a reversible cycle through which 1.00 mol of a monatomic ideal gas is taken. Volume
Question:
Figure shows a reversible cycle through which 1.00 mol of a monatomic ideal gas is taken. Volume Vc = 8.00%. Process bc is an adiabatic expansion, with pb = 10.0 atm and Vb = 1.00 x 10-3 m3. For the cycle, find
(a) The energy added to the gas as heat,
(b) The energy leaving the gas as heat,
(c) The net work done by the gas, and
(d) The efficiency of the cycle.
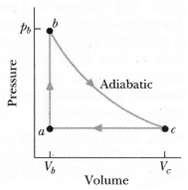
Transcribed Image Text:
Adiabatic V, V. Volume Pressure
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (14 reviews)
a Energy is added as heat during the portion of the process from a to b This portion occurs ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Question Posted:
Students also viewed these Thermodynamics questions
-
A 1.00-mol sample of a monatomic ideal gas is taken through the cycle shown in Figure P22.65. At point A, the pressure, volume, and temperature are Pi , Vi , and Ti , respectively. In terms of R and...
-
Suppose 1.00 mol of a monatomic ideal gas is taken from initial pressure p1 and volume V1 through two steps: (1) an isothermal expansion to volume 2.00V1 and (2) a pressure increase to 2.009 at...
-
The thermodynamic energy of a monatomic ideal gas is given by Find the partial derivatives and write the expression for dU using T ,V, and n as independent variables. Show that your differential is...
-
From our discussions of recent criticisms of research into cultural differences: Can we safely assume the existence of single national cultures? Is the influence of culture as an explanatory...
-
How can our trade theory of previous chapters be extended to incorporate changes in the nation's factor endowments, technology, and tastes? Is the resulting trade theory a dynamic theory of...
-
Consider the following hypothetical data for the U.S. economy in 2020 (all amounts are in trillions of dollars). Consumption ...................................... 11.0 Indirect business taxes...
-
9-2. En trminos de segmentos del mercado y productos, cules son las tres estrategias de segmentacin del mercado ?
-
Teradene Corporation purchased land as a factory site and contracted with Maxtor Construction to construct a factory. Teradene made the following expenditures related to the acquisition of the land,...
-
Instructions On March 31, 2099, the balances of the accounts appearing in the ledger of Royal Furnishings Company, a furniture store, are as follows Accounts Receivable $165 250 758,050 Accumulated...
-
a. How many Spanish-speaking operators and how many English-speaking operators does the hospital need to staff the call center during each two-hour shift of the day in order to answer all calls?...
-
A 500 W Carnot engine operates between constant temperature reservoirs at 100oC and 60.0oC. What is the rate at which energy is (a) Taken in by the engine as heat and (b) Exhausted by the engine as...
-
A Carnot engine is set up to produce a certain Work W per cycle. In each cycle, energy in the form of heat QH is transferred to the working substance of the engine from the higher-temperature thermal...
-
Story Corp was incorporated as a new business on January 1. The company is authorized to issue 20,000 shares of $5 par value common stock and 10,000 shares of 6%, $10 par value, cumulative,...
-
BREAD Products' pretax income for 2019 is * (1 Point) BREAD Products has no Work in Process or Finished Goods inventories at the close of business on December 31, 2018. The balances of BREAD's...
-
Convert the following line of code into assembly language. A (A B)+(BA) Where A and B are both 8-bit variables Activate Windows
-
14. Create a one variable Data Table from what you just copied and pasted giving the total sales for each department, and the Largest Sale from each department. Start your Criteria range in cell A1....
-
E4.1 (LO 1), C The following independent situations require professional judgment for determining when to recognize revenue from the transactions. a. Southwest Airlines sells you an advance-purchase...
-
Spring Flings Company, a fashion retailer that specializes in colorful graphic tees, prepares a master budget on a quarterly basis. The company has assembled the following data to assist in preparing...
-
Wetday is evaluating three projects: the Storm, the Cloud, and the Downpour. The companys cost of capital is 12%. These projects have the following cash flows. LO1 Storm Cloud Downpour Year 2 3 O...
-
As you rewrite these sentences, replace the cliches and buzzwords with plain language (if you don't recognize any of these terms, you can find definitions online): a. Being a jack-of-all-trades, Dave...
-
During a visit to the International Space Station, an astronaut was positioned motionless in the center of the station, out of reach of any solid object on which he could exert a force. Suggest a...
-
You have several identical balloons. You experimentally determine that a balloon will break if its volume exceeds 0.900 L. The pressure of the gas inside the balloon equals air pressure (1.00 atm....
-
A Jaguar XK8 convertible has an eight-cylinder engine. At the beginning of its compression stroke, one of the cylinders contains 499 cm3 of air at atmospheric pressure (1.01 X 105 Pa) and a...
-
A welder using a tank of volume 0.0750 m' fills it with oxygen (molar mass 32.0 g/mol) at a gauge pressure of 3.00 X 105 Pa and temperature of 37.0C. The tank has a small leak, and in time some of...
-
Bought an old van for 4000 from Peters promising to pay laterwhat is the transactions
-
Company has a following trade credit policy 1/10 N45. If you can borrow from a bank at 9,5% annual rate, would it be beneficial to borrow money and pay off invoices earlier?
-
Given the following exchange rates, which of the multiple-choice choices represents a potentially profitable inter-market arbitrage opportunity? 129.87/$1.1226/$0.00864/ 114.96/ B $0.8908/ (C)...

Study smarter with the SolutionInn App


