Question: How much work is required to set up the arrangement of Figure if q = 2.30pC, a = 64.0cm, and the particles are initially infinitely
How much work is required to set up the arrangement of Figure if q = 2.30pC, a = 64.0cm, and the particles are initially infinitely far apart and at rest?
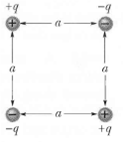
+9 +) +9
Step by Step Solution
3.35 Rating (164 Votes )
There are 3 Steps involved in it
We choose the zero of electric potential to be at infinity The initial electr... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
2-P-M-E-P (315).docx
120 KBs Word File


