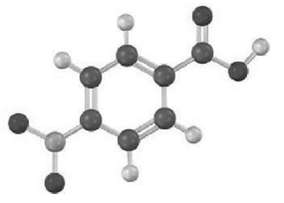
Question: How would you synthesize the following compound starting from benzene? More than one step isneeded.
How would you synthesize the following compound starting from benzene? More than one step isneeded.
Step by Step Solution
★★★★★
3.34 Rating (163 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

CH3CI AICI... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock
Document Format (1 attachment)
22-C-O-B (27).docx
120 KBs Word File


