In a set of experiments on a hypothetical one-electron atom, you measure the wavelengths of the photons
Question:
In a set of experiments on a hypothetical one-electron atom, you measure the wavelengths of the photons emitted from transitions ending in the ground state (n = I), as shown in the energy level diagram in Fig. 38.37. You also observe that it takes 17.50eV to ionize this atom.(a) What is the energy of the atom in each of the levels (n = I, n = 2, etc.) shown in the figure?(b) If an electron made a transition from the n = 4 to the n = 2 level what wavelength of light would itemit?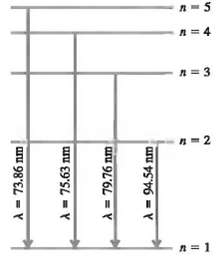
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Question Posted:





