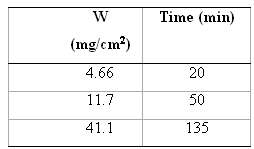
In the table, weight gain???time data for the oxidation of some metal at an elevated temperature are
Question:
In the table, weight gain???time data for the oxidation of some metal at an elevated temperature are tabulated.
(a) Determine whether the oxidation kinetics obey a linear, parabolic, or logarithmic rate expression.
(b) Now compute W after a time of 1000min.
Transcribed Image Text:
Time (min) (mg/cm?) 20 4.66 50 11.7 41.1 135
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
For this problem we are given weight gaintime data for the oxidation of some metal at an elevated t...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
In the table weight gain???time data for the oxidation of some metal at an elevated temperature are tabulated. (a) Determine whether the oxidation kinetics obey a linear, parabolic, or logarithmic...
-
In the table, weight gain-time data for the oxidation of copper at an elevated temperature are tabulated. W (mg/cm2) Time (min) 0.316........................... 15 0.524........................... 50...
-
For some transformation having kinetics that obey the Avrami equation (Equation 10.17), the parameter n is known to have a value of 1.7. If, after 100 s, the reaction is 50% complete, how long (total...
-
Two slot machines offer to double your money 3 times out of 5. Machine A takes $10 bets and Machine B takes $100 bets on each occasion. A risk-averse investor prefers to bet on A) Machine A B)...
-
1. The Wendt Corp had $10.0 Million in taxable income. (a) What is the company's federal income tax bill for the year? (b) Assume the firm receives an additional $1 million of interest income from...
-
What are the inventory classification for merchandising companies? What are the inventory categories typically used by manufacturing companies?
-
How does the book value of an asset being abandoned and replaced by a new asset affect the cash flow for purposes of computing the time-adjusted rate of return on investment? LO.1
-
Potential energy in the diamond structure (a) Show that for the diamond structure the Fourier component UG of the crystal potential seen by an electron is equal to zero for G = 2A, where A is a basis...
-
There are 4 stages in the life cycle of a company, and these are reflected in the Companys cash flows. What stage is characterized by low levels of operating cash flows and higher levels of cash...
-
Traditionally firms raise capital in the primary market issuing either fixed or floating rate debt. Both bond structures expose the firm to interest rate risk. The financial derivatives market...
-
In the table, weight gain-time data for the oxidation of copper at an elevated temperature are tabulated. (a) Determine whether the oxidation kinetics obey a linear, parabolic, or logarithmic rate...
-
A brine solution is used as a cooling medium in a steel heat exchanger. The brine is circulated within the heat exchanger and contains some dissolved oxygen. Suggest three methods, other than...
-
A 5.0 kg block moves in a straight line on a horizontal frictionless surface under the influence of a force that varies with position as shown in Figure. The scale of the figure's vertical axis is...
-
REQUIRED: Cost of production report under the following assumptions: Lost units - normal, discovered at the beginning Lost units - normal, discovered at the end Lost units - abnormal, discovered when...
-
ABC, Inc., manufactures only two products: Gadget A and Gadget B. The firm uses a single, plant wide overhead rate based on direct-labor hours. Production and product-costing data are as follows:...
-
.Jean Saburit has gone over the financial statements for Saburit Parts, Inc. The income statement has been prepared on an absorption costing basis and Saburit would like to have the statement revised...
-
When a constant force is applied to an object, the acceleration of the object varies inversely with its mass. When a certain constant force acts upon an object with mass 2 kg, the acceleration of the...
-
Use the following for all 3 circuits. V1 = 9.0 V, V = 12.0 V R = 2.0 ohms, R = 4.0 ohms, R3 = 6.0 ohms, R4 = 8.0 ohms C1 = 3.0 C = 3.0 (a) Find I in circuit A (b) Find I1 in circuit B R w R3 V R R4...
-
The balance in the unearned rent account for Jones Co. as of December 31 js $1,200. If Jones Co. failed to record the adjusting entry for $600 of rent earned during December, the effect on the...
-
Evaluate the integral, if it exists. Jo y(y + 1) dy
-
Provide a summary of customer balance characteristics for customers who made purchases. Include the minimum balance, maximum balance, and average balance, as shown in Figure P7.21. Figure P7.21...
-
What is the unique capability provided by pressure-to-pressure hydrostatic extrusion?
-
What types of rivets can be used when there is access to only one side of a joint?
-
Why might hubbing be an attractive way to produce a number of identical die cavities?
-
Duncan Inc. issued 500, $1,200, 8%, 25 year bonds on January 1, 2020, at 102. Interest is payable on January 1. Duncan uses straight-line amortization for bond discounts or premiums. INSTRUCTIONS:...
-
WISE-HOLLAND CORPORATION On June 15, 2013, Marianne Wise and Dory Holland came to your office for an initial meeting. The primary purpose of the meeting was to discuss Wise-Holland Corporation's tax...
-
Stock in ABC has a beta of 0.9. The market risk premium is 8%, and T-bills are currently yielding 5%. The company's most recent dividend is $1.60 per share, and dividends are expected to grow at a 6%...

Study smarter with the SolutionInn App


