The average speed (in meters per second) of a gas molecule is where T is the temperature
Question:
The average speed (in meters per second) of a gas molecule is
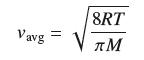
where T is the temperature (in kelvins), M is the molar mass (in kilograms per mole), and R = 8.31. Calculate dvavg/dT at T = 300 K for oxygen, which has a molar mass of 0.032 kg/mol.
Transcribed Image Text:
Vavg || 8RT ẨM
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
Using the form v av 8RTM 12 8RMT 12 ...View the full answer

Answered By

Felix Onchweri
I have enough knowledge to handle different assignments and projects in the computing world. Besides, I can handle essays in different fields such as business and history. I can also handle both short and long research issues as per the requirements of the client. I believe in early delivery of orders so that the client has enough time to go through the work before submitting it. Am indeed the best option that any client that can think about.
4.50+
5+ Reviews
19+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Mathematics questions
-
The graph shows speedometer readings, in meters per second (on the vertical axis), obtained as a skateboard travels along a straight-line path. How far does the board move between t = 3.00 s and t =...
-
Tempo Company's fixed budget (based on sales of 18,000 units) for the first quarter reveals the following. Fixed Budget Sales (18,000 units $210 per unit) $ 3,780,000 Cost of goods sold Direct...
-
The pedestrian walkway on the Golden Gate Bridge is about 75 m above the water below. This bridge is (unfortunately) a popular spot for some unhappy people, who attempt to jump off. Ignore air drag...
-
Define pricing practices of tesla INC as well as market structure.
-
When two mutually exclusive projects have different lives, how can an analyst determine which is better? What is the underlying assumption in this method?
-
Refer to Exercise 4.21. In Exercise 4.21 Required: 1. Calculate the global consumption ratios for the two products. 2. Using the activity consumption ratios for number of orders and number of setups,...
-
DEFAULT RISK PREMIUM The real risk-free rate, r*, is 2 5%. Inflation is expected to average 2 8% a year for the next 4 years, after which time inflation is expected to average 3 75% a year. Assume...
-
Treasury Stock Transactions The Stockholders Equity category of Little Joes balance sheet on January 1, 2010, appeared as follows: Common stock , $5 par, 40,000 shares issued and outstanding...
-
Problem 8-08 A company had $18 of sales per share for the year that just ended. You expect the company to grow their sales at 6.25 percent for the next five years. After that, you expect the company...
-
Show, using the limit definition of the derivative, that (x) = |x 2 4| is not differentiable at x = 2.
-
Compute the derivative of (x) = x 1/3 using the limit definition. Multiply the numerator and denominator in the difference quotient (x + h) (x) h by /2* + /1* / 1 (y + x) + /z(y + x)
-
Calvin is an executive on a business trip to the West Coast. He has driven his car on this trip and checks into the Hotel Ritz. The hotel has a guarded underground parking lot. Calvin gives his car...
-
Turn this information into an excel sheets with the excel formulas being shown P10.1 (LO 1) (Depreciation for Partial Period-SL, SYD, and DDB) Alladin Company purchased Machine #201 on May 1, 2025....
-
You are the Financial Analyst at Wellington Laboratories Ltd., a New Orleans, USA based bulk drugs manufacturer, which is evaluating the following project for manufacturing a new compound. Year Cash...
-
A variable mesh screen produces a linear and axisymmetric velocity profile as indicated below in the air flow through a 2-ft diameter circular cross section duct. The static pressures upstream and...
-
A vertical round steel rod 2 m long is securely held at its upper end. A weight can slide freely on the rod and its fall is arrested by a stop provided at the lower end of the rod. When the weight...
-
8) Determine the magnitudes of the forces F and P so that the single equivalent couple (i.e. the resultant of the three couples) acting on the triangular block is zero. Z -F F 3 m 10 N, 30 6 m 10 N 3...
-
The first picture in this Topic (the one that doesn't use determinants) shows a unique solution case. Produce a similar picture for the case of infinitely many solutions, and the case of no solutions.
-
A business had revenues of $280,000 and operating expenses of $315,000. Did the business (a) Incur a net loss (b) Realize net income?
-
Use a computer algebra system to find the exact area enclosed by the curves y = x5 - 6x3 + 4x and y = x.
-
Racing cars driven by Chris and Kelly are side by side at the start of a race. The table shows the velocities of each car (in miles per hour) during the first ten seconds of the race. Use the...
-
A cross-section of an airplane wing is shown. Measurements of the thickness of the wing, in centimeters, at 20-centimeter intervals are, 5.8, 20.3, 26.7, 29.0, 27.6, 27.3, 23.8, 20.5, 15.1, 8.7, and...
-
please help and show work thanks. Current Attempt in Progress Cullumber ProSystems needs a new signal conditioner module for a Large process control system it is designing. Current market conditions...
-
Required: 1. How many jobs must GTC average each month to break even? jobs per month 2. What is the operating income for GTC in a month with 89 jobs? Enter a net loss as a negative amount. $ What is...
-
Warren owns and manages a small business, which sells pet supplies to high street shops and market traders. Warren sells to customers on credit at 30 days credit terms. (Warren manages all aspects of...

Study smarter with the SolutionInn App


