A laser pointer used in a lecture hall emits light at 650 nm. What is the frequency
Question:
A laser pointer used in a lecture hall emits light at 650 nm. What is the frequency of this radiation? Using Figure 6.4, predict the color associated with this wavelength.
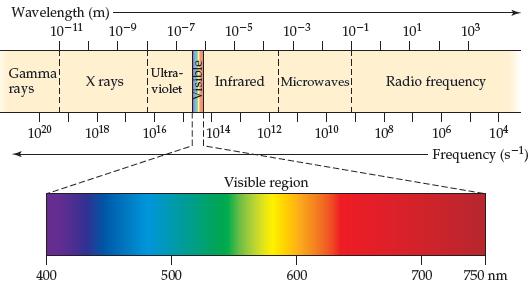
Wavelength (m)- 10-9 10-11 10-7 10-5 10-3 10-1 101 103 Gamma! X rays !Ultra- i violet rays Infrared iMicrowavesi Radio frequency 1020 1018 1016 1014 1012 1010 108 106 104 Frequency (s-1) Visible region 400 500 600 700 750 nm
Step by Step Answer:
The wavelength of a laser is 650 nm 650 10 ...View the full answer



Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Related Video
Using a few basic physics principles, you can impress your friends with a trick that makes a bottle disappear. For this experiment, you will need a mini plastic bottle, glycerin, a glass, and a funnel. First, open the bottle and pour some glycerin into it, then close the bottle tightly. Next, pour some water into the glass using a funnel. Place the mini bottle into the glass of water and it will look normal. This is because light travels through air faster than it travels through the glass and water, allowing our eyes to see the bottle inside the glass. However, when you fill the mini bottle with more glycerin, and pour glycerin into the glass, then put the glycerin filled bottle into the glass with glycerin in it. Half of the bottle that is submerged in the glycerin will become invisible as the light travels through glass and glycerin at the same speed, thus it does not bend and no refraction takes place, making the bottle invisible. This happens because both glass and glycerin have almost the same refractive index, which causes the speed of light to be the same in both mediums, causing no bending of light and making the bottle disappear.
Students also viewed these Sciences questions
-
(a) A red laser pointer emits light with a wavelength of 650 nm. What is the frequency of this light? (b) What is the energy of one of these photons? (c) The laser pointer emits light because...
-
(a) A green laser pointer emits light with a wavelength of 532 nm. What is the frequency of this light? (b) What is theenergy of one of these photons? (c) The laser pointer emits light because...
-
A laser used to read CDs emits red light of wavelength 700 nm. How many photons does it emit each second if its power is? (a) 0.10 W, (b) LOW?
-
Determine the force in member GC of the truss and state if this member is in tension or compression. Units Used: kip = 103 lb Given: F1 = 1000 lb F2 = 1000 lb F3 = 1000 lb F4 = 1000 lb a = 10 ft =...
-
y varies jointly as x and z and inversely as the square of w, and y = 12 / 5 when x = 16, z = 3, and w = 5 Find an equation of variation for the given situation.
-
An insulated mixing chamber receives 2 kg/s R-134a at 1 MPa, 100C in a line with low velocity. Another line with R-134a as saturated liquid 60C flows through a valve to the mixing...
-
4. Explain the difference between judicial restraint activism and judicial activism in contract law.
-
Austin, Inc., acquired 10 percent of McKenzie Corporation on January 1, 2014, for $210,000 although McKenzies book value on that date was $1,700,000. McKenzie held land that was undervalued by...
-
An entity with a higher proportion of fixed costs to total costs compared to an entity with a lower proportion of fixed to total costs is regarded as: a) less risky b) having a lower operating l...
-
When Justin and Nicole formed a partnership, Nicole signed over ownership of 1,000 shares of $15 par value stock she owns in a publicly traded corporation. The partners agreed to sell the stock in...
-
The energy from radiation can be used to cause the rupture of chemical bonds. A minimum energy of 242 kJ/mol is required to break the chlorinechlorine bond in Cl 2 . What is the longest wavelength of...
-
(a) Consider the following three statements: (i) A hydrogen atom in the n = 3 state can emit light at only two specific wavelengths, (ii) A hydrogen atom in the n = 2 state is at a lower energy than...
-
A monopole antenna extends vertically over a perfectly conducting plane, and has a linear current distribution. If the length of the antenna is 0.01, what value of I 0 is required to (a) Provide a...
-
A second-order system has effective mass 20 kg, spring constant 8 kN/m, damping coefficient 130 N s/m. It is subjected to harmonic input of 24 sin 15 t. Calculate the steady-state response of the...
-
A mail-order house uses 17,750 boxes a year. Carrying costs are 60 cents per box a year, and ordering costs are $96. The following price schedule applies. Number of BoxesPrice per Box1,000 to...
-
Terry Enterprises, Inc. has two divisions-the Foods division and the Clothes division. Historically, Terry has used the division's ROI as the performance measure for the bonus determinations. Terry...
-
Alpha is a retailer that has ten stores in New Orleans area that are supplied by 5 suppliers. It has box trucks for obtaining goods from its suppliers. The transportation manager in consultation with...
-
Submit quiz Jane was due to make loan payments of $1893 nine months ago, $4039 six month ago, and $883 in six months. Instead, she is to make a single payment today. If money is worth 7.6% and the...
-
The 2015 financial statements for Country Cousin Stores show total assets of $490,000, total liabilities of $290,000, net sales of $1,800,000, net income of $450,000, income from operations of...
-
B.) What is the approximate concentration of free Zn 2+ ion at equilibrium when 1.0010 -2 mol zinc nitrate is added to 1.00 L of a solution that is 1.080 M in OH - . For [Zn(OH) 4 ] 2- , K f = 4.610...
-
Three solutions are mixed together to form a single solution. One contains 0.2 mol Pb(CH3COO)2, the second contains 0.1 mol Na2S, and the third contains 0.1 mol CaCl2. (a) Write the net ionic...
-
Which of the following solutions has the largest concentration of solvated protons: (a) 0.2 M LiOH (b) 0.2 M HI (c) 1.0 M methyl alcohol (CH3OH)? Explain
-
Use the molecular representations shown here to classify each compound as either a nonelectrolyte, a weak electrolyte, or a strong electrolyte (see inside back cover for element color scheme). (a)...
-
A partial alphabetized list of adjusted account balances isshown for Dover Pacific Tours as at November 30, 2020 (all accountshave normal balances). Pat Dover, the owner, uses the followingaccount 2...
-
University Printers has two service departments (Maintenance and Personnel) and two operating departments (Printing and Developing). Management has decided to allocate maintenance costs on the basis...
-
Suny Sdn Bhd produces three products Z1, Z2 and Z3. Theseproducts are using the sametype of materials: D7, D8 and D9. Budgeted sales volumes of theseproducts for the year 2020are 2,500 units of Z1 2...

Study smarter with the SolutionInn App


