(a) Which of the following compounds, if any, is an ether? (b) Which compound, if any, is...
Question:
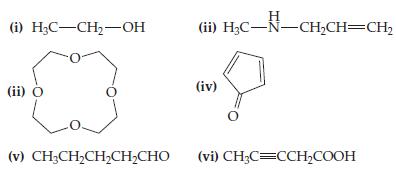
(a) Which of the following compounds, if any, is an ether?
(b) Which compound, if any, is an alcohol?
(c) Which compound, if any, would produce a basic solution if dissolved in water? (Assume solubility is not a problem).
(d) Which compound, if any, is a ketone?
(e) Which compound, if any, is an aldehyde?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Question Posted:





