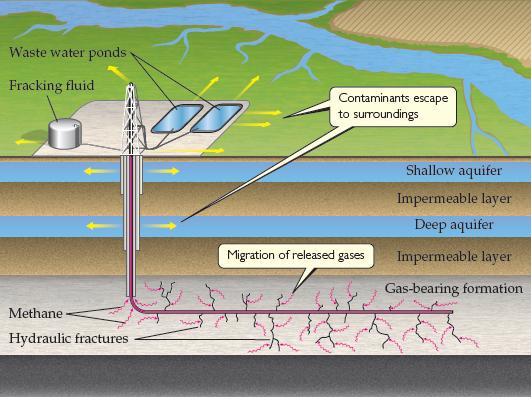
From study of Figure 18.22 describe the various ways in which operation of a fracking well site
Question:
From study of Figure 18.22 describe the various ways in which operation of a fracking well site could lead to environmental contamination.
Transcribed Image Text:
Waste water ponds Fracking fluid Contaminants escape to surroundings Shallow aquifer Impermeable layer Deep aquifer Migration of released gases Impermeable layer Gas-bearing formation Methane Hydraulic fractures
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
Firstly fracking leads to environmental contamination due to the large scale use ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Question Posted:
Students also viewed these Sciences questions
-
Describe the various ways in which the AICPAs Code is enforced.
-
Describe the various ways in which the Internet can facilitate generosity.
-
Figure depicts the various ways in which an average man gains and loess water in one day. One liter is ingested as food, and The body metabolically produces 0.3 L. In breathing air, the exchange is...
-
a. Determine IC and VCE for the network of Fig. 4.115. In Figure 4.115 b. Change β to 120 (50% increase), and determine the new values of lC and VCE for the network of Fig. 4.115. c....
-
What is meant by the moments of a PDF? What are the most frequently used moments?
-
At December 31, 2010, Reid Company had 600,000 shares of common stock issued and outstanding, 400,000 of which had been issued and outstanding throughout the year and 200,000 of which were issued on...
-
Differentiate the Fourier sine series by term in Problem 18. Show that the result is not the derivative of \(f(x)=x\). Data from Problem 18 An undamped, unforced Duffing Equation,...
-
The number of cars arriving per hour at Lundbergs Car Wash during the past 200 hours of operation is observed to be the following: NUMBER OF CARS ARRIVING FREQUENCY 3 or fewer ............. 0 4...
-
Please answer the question:6) What kind of decision should Twitter be making ?? rational,bounded rational or political? 2 answers
-
A hotel group prepares financial statements on a quarterly basis. The senior management is reviewing the performance of one hotel and making plans for next year. The managers have in front of them...
-
Where does the energy come from to evaporate the estimated 425,000 km 3 of water that annually leaves the oceans, as illustrated here? Water transport to the atmosphere Earth's atmosphere World ean
-
Which of the following reactions in the stratosphere cause an increase in temperature there? (a) O(g) + O2(g) O3 * (g) (b) O 3 * (g) + M(g) O 3 (g) + M * (g) (c) O 2 (g) + h 2O(g) (d) O(g) + N 2...
-
Use the following end-of-year price data to answer the following questions for the Harris and Pinwheel companies. Time ___________ Harris __________ Pinwheel 1 ........................$10...
-
Calculate each cost price. (4 marks) Rate of Mark-up on Cost Selling Price Price $135.00 15% $915.59 41.5% Calculate each cost price. (4 marks) Selling Price $16.79 $74.98 Cost Price Rate of Mark-up...
-
Use this definition with right endpoints to find an expression for the area under the graph of f as a limit. Do not evaluate the limit. f(x) = = 9 + sin(x), 0 X A = lim n 818 i = 1 Need Help? Submit...
-
Budgets are a basic management tool used to tie its planning activities to its operating activities. Budgets are important for communicating goals and expectations throughout the organization. In...
-
Explain the tax implications of deferred tax assets and liabilities with a focus on timing differences between book and tax income.
-
The U.S. airport system has evolved over the last 89 years into one of the most complex systems in the world. What are the roles of the FAA and the state in publicly owned and operated airports? In...
-
1. A housepainter needs to estimate the height of a condominium. He estimates the length of his shadow to be 7 feet long and the length of the building's shadow to be 42 feet long. Find the...
-
(8%) Problem 6: A student attaches a f= 3.5 kHz oscillator to one end of a metal rail of length L = 25 m. The student turns on the oscillator and uses a piezoelectric gauge at the other end to...
-
A 1.42-g sample of helium and an unknown mass of O2 are mixed in a flask at room temperature. The partial pressure of the helium is 42.5 torr, and that of the oxygen is 158 torr. What is the mass of...
-
A gaseous mixture of O2 and Kr has a density of 1.104 g/L at 355 torr and 400 K.What is the mole percent O2 in the mixture?
-
The density of a gas of unknown molar mass was measured as a function of pressure at 0oC, as in the table that follows. (a) Determine a precise molar mass for the gas. (b)Why is d/P not a constant as...
-
. According to the salary comparison in Figure 1, by the end of a 30-year career a college graduate is likely to earn: a. $22,895. b. $40,478. c. $686,850. bd. $1,214,340.
-
Miracle Works Inc. is located in Newfoundland. The company hires 3 eligible apprentices, paying each of them $30,000 in the current taxation year. It also acquires $2,000,000 in qualified depreciable...
-
Shareholders' equity and liabilities both have normal credit balances. Why are the shareholders' equity debit/credit rules more complex than liabilities? O The elements of Shareholders' Equity are...

Study smarter with the SolutionInn App


