Using the chain rule and stress transformation theory, develop the stressAiry function relations (7.6.7). Verify that this
Question:
Using the chain rule and stress transformation theory, develop the stress–Airy function relations (7.6.7). Verify that this form satisfies equilibrium identically.
Equation 7.6 .7
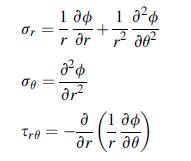
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Elasticity Theory Applications And Numerics
ISBN: 9780128159873
4th Edition
Authors: Martin H. Sadd Ph.D.
Question Posted:





