Question: A packed tower is used to remove component A from a gas stream mixture containing 12.0 mole% A and 88.0 mole% inerts. The gas mixture
A packed tower is used to remove component A from a gas stream mixture containing 12.0 mole% A and 88.0 mole% inerts. The gas mixture is fed to the bottom of the tower, and the desired superficial molar velocity of the gas stream into the tower (G1) is 5.0 kgmole/m2· h. Solvent containing no solute A is fed into the top of the tower. The desired composition of solute A exiting with the liquid is 3.0 mole%, and the desired composition of solute A exiting with the gas (yA2) is 1.0 mole%. Through independent studies, overall average mass-transfer coefficient based on the gas phase for this tower at the desired superficial molar velocities was determined to be KGα = 1.96 kgmole/m3· h · atm. The process is carried out at 20°C and 15 psia total system pressure. Equilibrium distribution data for solute A in the solvent at 20°C and 15 psia total system pressure is provided in the table below.
a. Specify the molar flow rate and mole fraction composition of all terminal streams in the process. What is the superficial molar velocity of solvent into the tower, L2?
b. Plot out the equilibrium line and operating line in mole ratio coordinates.
c. What is the packed height of the tower required to accomplish the separation?
Equilibrium distribution data, in mole fraction coordinates at 20°C and 15 psia total system pressure:
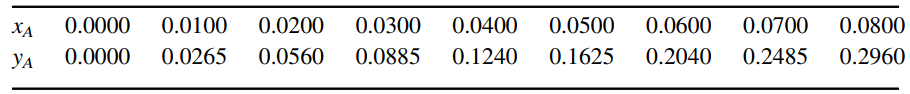
0.0400 0.0800 0.0000 0.0100 0.0200 0.0300 0.0500 0.0600 0.0700 XA 0.1240 0.1625 0.0000 0.0265 0.0560 0.0885 0.2040 0.2485 0.2960
Step by Step Solution
3.50 Rating (167 Votes )
There are 3 Steps involved in it
a Determine L 2 and mole fraction compositions of terminal streams ... View full answer

Get step-by-step solutions from verified subject matter experts


